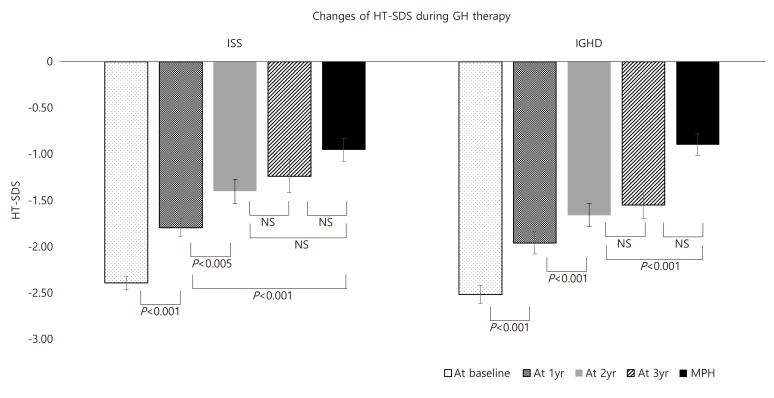
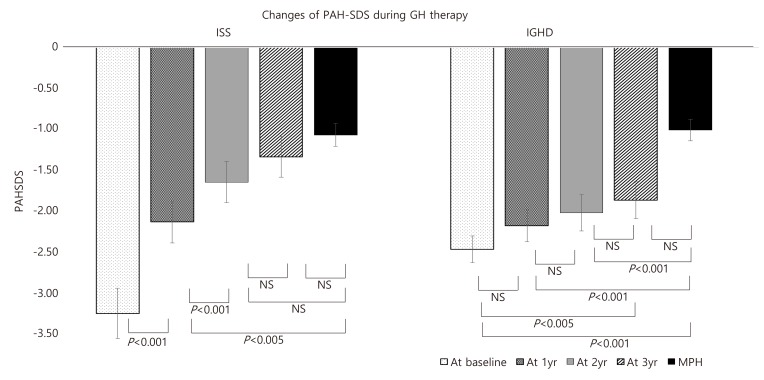
1. Korean Society of Pediatric Endocrinology. Pediatric Endocrinology. 3rd ed. Seoul: Gunja Co.. 2014;pp 83.
2. Ranke MB. Towards a consensus on the definition of idiopathic short stature. Horm Res 1996;45(Suppl 2):64ŌĆō66. PMID:
8805048.


3. Cohen P, Rogol AD, Deal CL, Saenger P, Reiter EO, Ross JL, et al. Consensus statement on the diagnosis and treatment of children with idiopathic short stature: a summary of the Growth Hormone Research Society, the Lawson Wilkins Pediatric Endocrine Society, and the European Society for Paediatric Endocrinology Workshop. J Clin Endocrinol Metab 2008;93:4210ŌĆō4217. PMID:
18782877.



4. Loche S, Carta L, Ibba A, Guzzetti C. Growth hormone treatment in non-growth hormone-deficient children. Ann Pediatr Endocrinol Metab 2014;19:1ŌĆō7. PMID:
24926456.



5. Gubitosi-Klug RA, Cuttler L. Idiopathic short stature. Endocrinol Metab Clin North Am 2005;34:565ŌĆō580. viiiPMID:
16085160.


6. Kim DH, Shin HJ, Chung SC, Park MJ. Response of growth hormone treatment to final height in children with growth hormone deficiency and familial short stature. J Korean Soc Pediatr Endocrinol 1999;4:159ŌĆō169.
7. Kang JC, Choi YS, Choi IK, Kim HS, Kim DH. The effect of growth hormone on patients with growth hormone deficiency and idiopathic short stature. Korean J Pediatr 2004;47:310ŌĆō318.
8. Kim SA, Choe YR, Yang EM, Kim CJ. Comparison of growth hormone treatment in patients with idiopathic short stature and idiopathic growth hormone deficiency. Chonnam Med J 2014;50:63ŌĆō66. PMID:
25229018.



9. Greulich WW, Pyle SI. Radiographic atlas of skeletal development of the hand and wrist. 2nd ed. Stanford: Stanford University Press. 1959.
10. Bayley N, Pinneau SR. Tables for predicting adult height from skeletal age: revised for use with the Greulich-Pyle hand standards. J Pediatr 1952;40:423ŌĆō441. PMID:
14918032.


11. Moon JS, Lee SY, Nam CM, Choi JM, Choe BK, Seo JW, et al. 2007 Korean National Growth Charts: review of developmental process and an outlook. Korean J Pediatr 2008;51:1ŌĆō25.

12. Hyun SE, Lee BC, Suh BK, Chung SC, Ko CW, Kim HS, et al. Reference values for serum levels of insulin-like growth factor-I and insulin-like growth factor binding protein-3 in Korean children and adolescents. Clin Biochem 2012;45:16ŌĆō21. PMID:
22032863.


13. Growth Hormone Research Society. Consensus guidelines for the diagnosis and treatment of growth hormone (GH) deficiency in childhood and adolescence: summary statement of the GH Research Society. GH Research Society. J Clin Endocrinol Metab 2000;85:3990ŌĆō3993. PMID:
11095419.

14. Savage MO, Bang P. The variability of responses to growth hormone therapy in children with short stature. Indian J Endocrinol Metab 2012;16(Suppl 2):S178ŌĆōS184. PMID:
23565373.



15. Lee BC, Lee SJ, Jung MH. Final height in growth hormone deficient children treated with growth hormone. J Korean Soc Pediatr Endocrinol 2003;8:27ŌĆō33.
16. Choi IJ, Hwang JS, Shin CH, Yang SW. Factors affecting on final adult height and total height gain in children with idiopathic and organic growth hormone deficiency after growth hormone treatment. J Korean Pediatr Soc 2003;46:803ŌĆō810.
17. Kim HS, Yang SW, Yoo HW, Suh BK, Ko CW, Chung WY, et al. Efficacy of short-term growth hormone treatment in prepubertal children with idiopathic short stature. Yonsei Med J 2014;55:53ŌĆō60. PMID:
24339287.



18. Leschek EW, Rose SR, Yanovski JA, Troendle JF, Quigley CA, Chipman JJ, et al. Effect of growth hormone treatment on adult height in peripubertal children with idiopathic short stature: a randomized, double-blind, placebo-controlled trial. J Clin Endocrinol Metab 2004;89:3140ŌĆō3148. PMID:
15240584.


19. Albertsson-Wikland K, Aronson AS, Gustafsson J, Hagen├żs L, Ivarsson SA, Jonsson B, et al. Dose-dependent effect of growth hormone on final height in children with short stature without growth hormone deficiency. J Clin Endocrinol Metab 2008;93:4342ŌĆō4350. PMID:
18728172.



20. McCaughey ES, Mulligan J, Voss LD, Betts PR. Randomised trial of growth hormone in short normal girls. Lancet 1998;351:940ŌĆō944. PMID:
9734940.


21. Cohen P, Weng W, Rogol AD, Rosenfeld RG, Kappelgaard AM, Germak J. Dose-sparing and safety-enhancing effects of an IGF-I-based dosing regimen in short children treated with growth hormone in a 2-year randomized controlled trial: therapeutic and pharmacoeconomic considerations. Clin Endocrinol (Oxf) 2014;81:71ŌĆō76. PMID:
24428305.



22. Cohen P, Germak J, Rogol AD, Weng W, Kappelgaard AM, Rosenfeld RG, et al. Variable degree of growth hormone (GH) and insulin-like growth factor (IGF) sensitivity in children with idiopathic short stature compared with GH-deficient patients: evidence from an IGF-based dosing study of short children. J Clin Endocrinol Metab 2010;95:2089ŌĆō2098. PMID:
20207829.



23. Wit JM, Rekers-Mombarg LT, Cutler GB, Crowe B, Beck TJ, Roberts K, et al. Growth hormone (GH) treatment to final height in children with idiopathic short stature: evidence for a dose effect. J Pediatr 2005;146:45ŌĆō53. PMID:
15644821.


24. Sotos JF, Tokar NJ. Growth hormone significantly increases the adult height of children with idiopathic short stature: comparison of subgroups and benefit. Int J Pediatr Endocrinol 2014;2014:15. PMID:
25075207.



25. Park P, Cohen P. The role of insulin-like growth factor I monitoring in growth hormone-treated children. Horm Res 2004;62(Suppl 1):59ŌĆō65. PMID:
15761234.