 |
 |
- Search
| Ann Pediatr Endocrinol Metab > Volume 28(2); 2023 > Article |
|
Abstract
Purpose
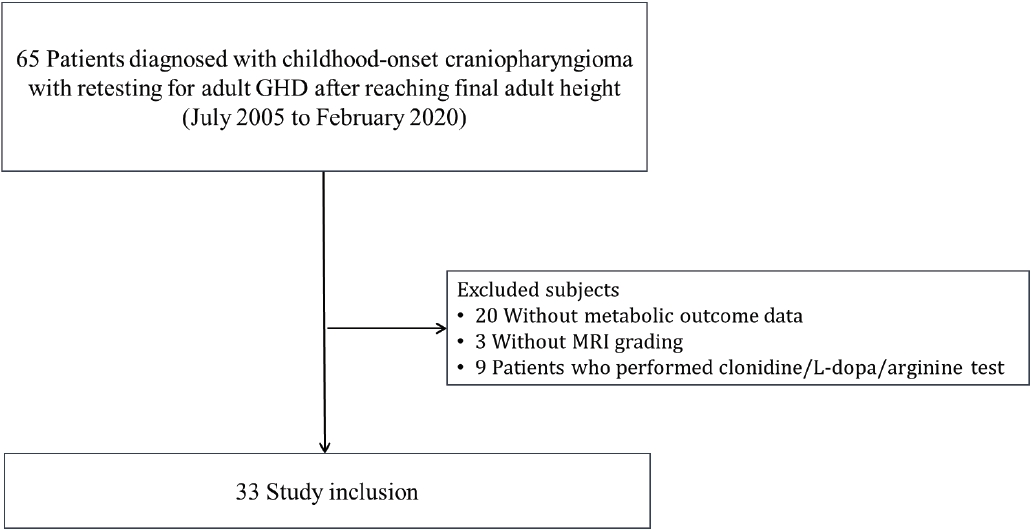
Methods
Results
Notes
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Fig.┬Ā1.
Table┬Ā1.
| Characteristic | Value |
|---|---|
| Male sex | 17 (51.5) |
| Age at initial operation (yr) | 7.6┬▒3.7 |
| Hypothalamic involvement | |
| ŌĆāNo | 3 (9.1) |
| ŌĆāMild | 14 (42.4) |
| ŌĆāExtensive | 16 (48.5) |
| Radiation therapy history | 6 (18.2) |
| Recurrence history | 10 (30.3) |
| Family history of cardiometabolic disease | 22 (66.7) |
| Hormone deficiency | |
| ŌĆāGonadotropins | 32 (97.0) |
| ŌĆāACTH | 32 (97.0) |
| ŌĆāTSH | 33 (100) |
| ŌĆāADH | 28 (84.8) |
| GH treatment | |
| ŌĆāAge at the start of childhood GH therapy (yr) | 10.0┬▒3.6 |
| ŌĆāDuration of childhood GH therapy (yr) | 5.5┬▒3.4 |
| ŌĆāAge at the time of GH discontinuation (yr) | 15.3┬▒3.1 |
| ŌĆāDuration off GH therapy before GH retesting (yr) | 2.1┬▒1.7 |
| ŌĆāDuration off GH therapy (mo) | |
| ŌĆāŌĆā<6 | 6 (18.2) |
| ŌĆāŌĆāŌēź6 | 27 (81.8) |
| At the time of retesting | |
| ŌĆāAge at retesting (yr) | 17.7┬▒2.5 |
| ŌĆāHeight z-score | 0.5┬▒0.9 |
| ŌĆāWeight z-score | 0.9┬▒1.3 |
| ŌĆāBody mass index z-score | 0.8┬▒1.5 |
| ŌĆāObesity | 9 (27.3) |
| ŌĆāCentral obesityŌĆĀ | 11 (42.3) |
| ŌĆāSBP Ōēź 130 mmHg or DBP Ōēź 85 mmHg | 3 (9.1) |
| ŌĆāFasting glucose Ōēź 100 (mg/dL) | 3 (9.1) |
| ŌĆāTriglycerides Ōēź 150 (mg/dL) | 15 (45.5) |
| ŌĆāHDL-C < 40 mg/dL (male) or < 50 mg/dL (female) | 19 (57.6) |
| ŌĆāMetabolic syndromeŌĆĀ,ŌĆĪ | 4 (15.4) |
Table┬Ā2.
| Characteristic |
HI |
Post-GH discontinuation duration |
|||
|---|---|---|---|---|---|
| No or mild (n=17) | Extensive (n=16) | <6 mo (n=6) | Ōēź6 mo (n=27) | ||
| Male sex | 10 (58.8) | 8 (50.0) | 3 (50.0) | 15 (55.6) | |
| Age at initial operation (yr) | 6.7┬▒4.5 | 8.6┬▒2.5 | 9.3┬▒3.9 | 7.3┬▒3.7 | |
| Radiation therapy history | 3 (17.6) | 3 (18.8) | 1 (16.7) | 5 (18.5) | |
| Recurrence history | 4 (23.5) | 6 (37.5) | 1 (16.7) | 9 (33.3) | |
| Family history of cardiometabolic disease | 9 (52.9) | 13 (81.3) | 5 (83.3) | 17 (63.0) | |
| Hormone deficiency | |||||
| Gonadotropins | 17 (100) | 15 (93.8) | 5 (83.3) | 27 (100) | |
| ACTH | 17 (100) | 15 (93.8) | 5 (83.3) | 27 (100) | |
| TSH | 17 (100) | 16 (100) | 6 (100) | 27 (100) | |
| ADH | 12* (70.6) | 16* (100) | 6 (100) | 22 (81.5) | |
| GH treatment | |||||
| Age at the start of childhood GH therapy (yr) | 9.1┬▒4.2 | 10.9┬▒2.5 | 11.8┬▒3.3 | 9.6┬▒3.5 | |
| Duration of childhood GH therapy (yr) | 6.8┬▒3.4* | 4.2┬▒2.8* | 5.0┬▒3.1 | 5.8┬▒3.4 | |
| Age at the time of GH discontinuation (yr) | 16.0┬▒3.0 | 14.6┬▒3.1 | 15.7┬▒1.7 | 15.2┬▒3.3 | |
| GH discontinuation duration (yr) | 1.9┬▒1.3 | 2.4┬▒2.1 | 0.2┬▒0.2* | 2.6┬▒1.6* | |
| At the time of GH retesting | |||||
| Age at evaluation (yr) | 17.9┬▒2.5 | 17.4┬▒2.6 | 15.9┬▒1.7* | 18.1┬▒2.5* | |
| Postoperative duration (yr) | 11.2┬▒3.5* | 8.8┬▒3.0* | 6.6┬▒3.3* | 10.8┬▒3.0* | |
| Height z-score | 0.3┬▒0.8 | 0.7┬▒1.0 | 0.5┬▒1.0 | 0.5┬▒0.9 | |
| Weight z-score | 0.3┬▒1.2* | 1.6┬▒1.2* | 0.6┬▒1.6 | 1.0┬▒1.3 | |
| Body mass index z-score | 0.2┬▒1.3* | 1.4┬▒1.5* | 0.4┬▒2.0 | 0.9┬▒1.4 | |
| Obesity | 2 (11.8) | 7 (43.8) | 1 (16.7) | 8 (29.6) | |
| Waist circumference (cm)ŌĆĀ | 82.3┬▒9.5 | 88.1┬▒14.7 | 82.6┬▒20.3 | 85.2┬▒10.7 | |
| Central obesityŌĆĀ | 5 (33.3) | 6 (54.5) | 1 (25.0) | 10 (45.5) | |
| Fasting glucose Ōēź 100 (mg/dL) | 1 (5.9) | 2 (12.5) | 0 (0.0) | 3 (9.1) | |
| Triglycerides Ōēź 150 (mg/dL) | 7 (41.2) | 8 (50.0) | 1 (16.7) | 14 (51.9) | |
| HDL-C < 40 mg/dL (male) or <50 mg/dL (female) | 8 (47.1) | 11 (68.8) | 1 (16.7) | 18 (66.7) | |
| SBP Ōēź 130 mmHg or DBP Ōēź 85 mmHg | 1 (5.9) | 2 (12.5) | 0 (0.0) | 3 (11.1) | |
| Metabolic syndromeŌĆĀ,ŌĆĪ | 1 (6.7) | 3 (27.3) | 0 (0.0) | 4 (18.2) | |
Table┬Ā3.
| Variable | Total |
Hypothalamic involvement |
Post-GH discontinuation duration |
Two-way ANOVA (F-values) |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No or mild (n=17) | Extensive (n=16) | Effect sizeŌĆĀ | <6 mo (n=6) | Ōēź6 mo (n=27) | Effect sizeŌĆĀ | HI | Discontinuation duration | HI ├Ś Discontinuation duration | ||
| BMI (z-score) | 0.8┬▒1.5 | 0.2┬▒1.3 | 1.4┬▒1.5* | 0.162 | 0.4┬▒2.0 | 0.9┬▒1.4 | 0.017 | 5.25* | 0.66 | 0.43 |
| FBS (mg/dL) | 84.1┬▒9.7 | 83.5┬▒7.9 | 84.8┬▒11.6 | 0.004 | 82.3┬▒8.2 | 84.5┬▒10.2 | 0.007 | 0.47 | 0.22 | 0.40 |
| TGŌĆĪ (mg/dL) | 116.0 (78.5ŌĆō203.0) | 111.0 (68.0ŌĆō184.0) | 146.5 (90.8ŌĆō208.8) | 0.011 | 77.5 (61.5ŌĆō135.8) | 156.0 (90.0ŌĆō208.0) | 0.080 | 0.51 | 2.60 | 0.15 |
| HDL-C (mg/dL) | 42.1┬▒12.8 | 45.2┬▒15.0 | 38.8┬▒9.3 | 0.048 | 51.8┬▒22.6 | 39.9┬▒8.7** | 0.133 | 7.96* | 5.78* | 5.49* |
| Fasting insulinŌĆĪ (╬╝IU/mL) | 9.1 (6.0ŌĆō15.0) | 7.0 (4.7ŌĆō11.3) | 13.2 (6.9ŌĆō15.9) | 0.074 | 6.6 (6.0ŌĆō10.0) | 10.9 (5.5ŌĆō15.3) | 0.030 | 1.92 | 1.02 | 0.08 |
| HOMA-IRŌĆĪ | 1.9 (1.2ŌĆō3.4) | 1.6 (1.0ŌĆō2.5) | 2.7 (1.4ŌĆō3.7) | 0.073 | 1.4 (1.1ŌĆō2.1) | 2.2 (1.2ŌĆō3.5) | 0.033 | 2.13 | 1.11 | 0.16 |
| SBP (mmHg) | 112.9┬▒9.2 | 112.6┬▒8.3 | 113.3┬▒10.4 | 0.001 | 108.3┬▒9.9 | 114.0┬▒9.0 | 0.057 | 0.83 | 1.82 | 1.39 |
| DBP (mmHg) | 67.5┬▒9.3 | 65.5┬▒8.6 | 69.7┬▒9.9 | 0.052 | 69.2┬▒6.8 | 67.1┬▒9.9 | 0.007 | 1.10 | 0.21 | 0.01 |
Values are presented as mean┬▒standard deviation or median (interquartile range).
GH, growth hormone; ANOVA, analysis of variance; HI, hypothalamic involvement; BMI, body mass index; FBS, fasting blood sugar; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment of insulin resistance. SBP, systolic blood pressure; DBP, diastolic blood pressure
Table┬Ā4.
| Variable | BMI (z-score) | FBS (mg/dL) | TGŌĆĀ (mg/dL) | HDL-C (mg/dL) | Fasting insulinŌĆĀ (╬╝IU/mL) | HOMA-IRŌĆĀ | SBP (mmHg) | DBP (mmHg) |
|---|---|---|---|---|---|---|---|---|
| Extensive HI (vs. no or mild) | 1.0 (-0.2 to 2.2) | -0.8 (-9.2 to 7.6) | 0.1 (-0.4 to 0.7) | -13.3* (-25.2 to -1.5) | 0.3 (-0.3 to 0.8) | 0.3 (-0.3 to 0.8) | 1.3 (-4.6 to 7.2) | 3.7 (-4.6 to 11.4) |
| Ōēź6 mo post-GH-discontinuation (vs. <6 mo) | 0.0 (-2.0 to 2.0) | 5.8 (-8.1 to 19.6) | 0.6 (-0.2 to 1.4) | -17.4* (-33.3 to -1.6) | 0.1 (-0.8 to 1.0) | 0.1 (-0.8 to 1.0) | 0.8 (-8.9 to 10.5) | -3.8 (-16.5 to 9.0) |
| Duration of childhood GH therapy | -0.2 (-0.6 to 0.1) | 0.3 (-2.0 to 2.6) | 0.0 (-0.2 to 0.1) | 1.1 (-1.9 to 4.1) | -0.1 (-0.2 to 0.1) | -0.1 (-0.2 to 0.1) | -1.9 (-3.7 to -0.2) | -0.4 (-2.4 to 1.7) |
| Age at the start of childhood GH therapy | -0.1 (-0.5 to 0.3) | 0.4 (-2.3 to 3.1) | 0.0 (-0.2 to 0.2) | 0.4 (-3.1 to 3.9) | 0.0 (-0.2 to 0.2) | 0.0 (-0.2 to 0.2) | 0.7 (-1.3 to 2.8) | 1.7 (-0.7 to 4.0) |
| Age at evaluation | 0.2 (-0.3 to 0.6) | -0.1 (-3.0 to 2.8) | 0.0 (-0.2 to 0.2) | 0.3 (-3.5 to 4.0) | 0.1 (-0.1 to 0.2) | 0.1 (-0.1 to 0.2) | -0.7 (-2.9 to 1.5) | -1.2 (-3.7 to 1.4) |
| Male (vs. female) | -0.5 (-1.6 to 0.6) | -2.1 (-9.8 to 5.5) | 0.3 (-0.2 to 0.9) | 3.2 (-5.8 to 12.2) | -0.1 (-0.5 to 0.4) | -0.1 (-0.6 to 0.4) | -8.4* (-13.8 to -3.0) | -3.1 (-10.2 to 4.0) |
| Postoperative duration | 0.1 (-0.2 to 0.3) | -0.1 (-2.1 to 1.9) | 0.1 (-0.1 to 0.2) | -0.9 (-3.5 to 1.7) | 0.1 (-0.1 to 0.2) | 0.1 (-0.1 to 0.2) | 2.7* (1.2 to 4.2) | 1.4 (-0.4 to 3.2) |
| Family historyŌĆĪ (vs. no history) | 0.6 (-0.6 to 1.9) | 8.5 (-0.2 to 17.2) | -0.2 (-0.7 to 0.3) | -0.2 (-0.8 to 0.4) | 0.3 (-0.2 to 0.9) | 0.4 (-0.1 to 1.0) | 1.7 (-4.4 to 7.8) | 4.2 (-3.9 to 12.2) |
| R2 | 0.283 | 0.187 | 0.227 | 0.411 | 0.252 | 0.279 | 0.554 | 0.249 |
Values are presented as mean difference (95% confidence interval).
ANCOVA, analysis of covariance; GH, growth hormone; HI, hypothalamic involvement; BMI, body mass index; FBS, fasting blood sugar; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment of insulin resistance. SBP, systolic blood pressure; DBP, diastolic blood pressure.
References
-
METRICS

-
- 0 Crossref
- 1 Scopus
- 2,379 View
- 140 Download
- Related articles in APEM






