 |
 |
- Search
| Ann Pediatr Endocrinol Metab > Volume 28(2); 2023 > Article |
|
This article has been corrected. See Ann Pediatr Endocrinol Metab. 2023 Sep 19; 28(3): 231.
Abstract
Thyroid hormone plays a vital role in regulating human metabolism. They affect the functions of major organs, such as the brain, liver, skeletal muscle, and heart. Hypothyroidism can lead to dilated cardiomyopathy and decreased heart function. In this report, we describe a case of a teenage boy who developed dilated cardiomyopathy due to hypothyroidism and was considered to undergo heart transplantation. Levothyroxine monotherapy was initiated but produced no improvement. Thereafter, a combination therapy of liothyronine and levothyroxine was administered, and heart function was gradually restored; he recovered completely after 6 months. Cardiac myocytes respond more specifically to liothyronine than to levothyroxine. Therefore, we suggest that liothyronine and levothyroxine combination therapy should be considered rather than levothyroxine monotherapy for hypothyroidism accompanied by heart disease.
┬Ę Thyroid hormone plays an important role in regulating human metabolism and can also affect the heart and vascular system. This report presents a case of successful treatment with combination therapy of T3 and T4 in a teenage boy with hypothyroidism-induced dilated cardiomyopathy.
Thyroid hormone (TH) regulates metabolism, particularly metabolic processes essential for normal growth and development [1]. TH-mediated metabolic control affects the brain, liver, and pancreas as well as white fat tissue, brown fat tissue, and skeletal muscle [2]. Furthermore, TH has many important effects on the heart and vascular system. Cellular mechanisms of TH alter liothyronine (T3)-regulated gene expression leading to changes in cardiac contractility, calcium cycling, and diastolic relaxation of the myocardium [3,4]. In addition, TH has nongenomic effects on cardiac myocytes, which are also T3 hormone-mediated effects [5]. Therefore, hypothyroidism may increase systemic vascular resistance, further contributing to a decrease in cardiac output, increased myocardial stiffness, and left ventricular diastolic dysfunction [3]. It can even lead to dilated cardiomyopathy (DCMP), which requires immediate TH stimulation. Cardiac myocytes appear to transport T3 in a marked preference for levothyroxine (T4) and are extremely sensitive to changes in serum T3 concentration. Therefore, T3 supplementation should be considered for hypothyroidism- induced DCMP [6,7]. We report a case of a teenage boy who developed DCMP due to hypothyroidism. His condition did not improve with T4 monotherapy but remarkably improved with T3 and T4 combination therapy.
A 16-year-old boy was referred to Severance Children's Hospital for aggressive exacerbation of DCMP. Before being transferred, he presented with New York Heart Association functional class IIIŌĆōIV dyspnea at the hospital. The cardiac enzyme showed elevated serum levels of N-terminal pro-brain natriuretic peptide 4,965 pg/mL (reference range, <153 pg/mL), creatine kinase 4,627 IU/L (reference range, 20ŌĆō200 IU/L), and creatine kinase myocardial band 25.6 ng/mL (reference range, 0ŌĆō5 mg/mL). He presented with a dilated left ventricular cavity with a diastolic dimension of 78 mm and decreased global systolic function with an ejection fraction of 43% on echocardiography. His condition was diagnosed as DCMP with congestive heart failure, and furosemide and spironolactone therapy were initiated.
Despite initiating medications for heart failure, his symptoms worsened, and further evaluation was performed for a differential diagnosis. Thyroid function test performed on the 10th day of admission showed hypothyroidism with levels of free T4 at 0.06 ng/dL (reference range, 0.80ŌĆō1.90 ng/dL), T3 at 48 ng/dL (reference range, 78ŌĆō182 ng/dL), and thyroid-stimulating hormone (TSH) at 98 ╬╝IU/mL (reference range, 0.17ŌĆō4.85 ╬╝IU/mL). The antimicrosomal antibody and TSH receptor antibody levels were elevated at 704 U/mL (reference range, 0ŌĆō60 U/mL) and >400 IU/L (positive, >1.5 IU/L), respectively, but thyroglobulin antibody and thyroid-stimulating antibody were negative. On thyroid ultrasonography, the patient had lobular-contoured homogeneous echogenicity of both thyroid glands. Based on these findings, he was started on levothyroxine 150 ╬╝g/day monotherapy (1.5 ╬╝g/kg). However, his cardiac function and general condition worsened, and he was transferred to our hospital on the 40th day.
On physical examination after transfer, the patient had bilateral non-pitting edema of the foot. Cardiovascular investigation revealed tachycardia with no murmurs or gallops. His height and body weight were 163 cm and 93.8 kg, respectively, and his vital signs were as follows: blood pressure, 89/44 mmHg; heart rate, 105 beats/min; respiratory rate, 27 breaths/min; and O2 saturation, 95% in room air. Chest x-ray showed cardiomegaly and pleural effusion (Fig. 1). Electrocardiography demonstrated sinus tachycardia (Fig. 2). The dilated left ventricular diastolic dimension was 74 mm, with an ejection fraction of 26% on echocardiography. Heart magnetic resonance imaging showed increased left ventricular end-diastolic volume (LVEDV) and decreased left ventricular ejection fraction (LVEF) (LVEDV 340.56 mL, LVEF 25.48%). He underwent cardiac biopsy, viral studies, inflammatory marker analysis, and genetic tests to identify the cause of DCMP, but the results for all were negative. He was administered enalapril, carvedilol, and ivabradine for heart failure and was considered a candidate for heart transplantation.
Although he took 150 ╬╝g of T4 for 40 days, TFT results revealed a high TSH (26.33 ╬╝IU/mL), low T3 (<0.40 ng/mL), and normal free T4 (1.27 ng/dL) levels on the first day after transfer, after which he was referred to our department. We assumed that the cause of DCMP was hypothyroidism; therefore, T3 supplementation was essential. Administration of 12.5 ╬╝g of T3 and 150 ╬╝g of T4 was initiated. However, after 3 days, the condition worsened, and additional administration of T3 12.5 ╬╝g and T4 50 ╬╝g was started. His condition and heart function slowly improved; after 2 weeks of T3 and T4 administration, the TSH (11.60 ╬╝IU/mL) level was mildly elevated, but the T3 (0.97 ng/mL) and free T4 (1.02 ng/dL) levels were within normal ranges. Thereafter, we additionally administered 6.25 ╬╝g of T3 and 25 ╬╝g of T4 to achieve a normal TSH level. At 6 weeks after T3 and T4 administration, his heart function had noticeably recovered with a left ventricular end-diastolic diameter of 71.5 mm and LVEF of 35%, and his weight had decreased from 93.8 kg to 71.7 kg without pitting edema. At that time, although there were no symptoms of hyperthyroidism, TFT showed mild hyperthyroidism with suppressed TSH (0.05 ╬╝IU/mL), elevated T3 (2.55 ng/mL), and normal free T4 (1.08 ng/dL) levels. Therefore, we adjusted the T3 and T4 doses to 25 ╬╝g and 200 ╬╝g, respectively, and he was discharged.
During follow-up, the patient showed a gradual decrease in the volume of the left ventricular chamber with a diastolic dimension of 61 mm at one year of discharge, and his left ventricular systolic function had returned to normal with an ejection fraction of 62%. We advised him to stop T3 administration 2 months after discharge; the TFT was stable with T4 administration alone, and the patient had no symptoms of hypothyroidism. Follow-up thyroid function and echocardiography findings are shown in Table 1 and Fig. 3, respectively. Cardiac medication was continued with enalapril and carvedilol in small amounts. He was advised to take only 150 ╬╝g of T4, and his heart function remained normal for 3 years.
This study was approved by the Institutional Review Board of Yonsei University Severance Hospital (approval number: 4-2021-1173). Informed consent was obtained from the patient and his guardian for publication of this case report.
TH is essential for normal development, growth, neural differentiation, and metabolic regulation [1]. Hypothyroidism is associated with hypometabolism characterized by reduced resting energy expend iture, weight gain, increased cholesterol levels, reduced lipolysis, and reduced gluconeogenesis [8]. Because TH has an important effect on the cardiovascular system, hypothyroidism might lead to DCMP. Wang et al. [9] reported that low thyroid function could lead to cardiac atrophy with chamber dilatation, impaired myocardial blood flow, loss of arterioles, and severe systolic dysfuncti on, resembling the pathological changes in DCMP. DCMP is characterized by an enlarged and poorly contractile left ventricular, is usually irreversible, and progresses to heart failure [10]. Bezdah et al. [11] reported a case of a patient with DCMP and heart failure due to severe hypothyroidism, who has recovered to euthyroidism with T4 therapy. They suggested that hypothyroidism should be considered as a trigger when DCMP is diagnosed.
Our patient had no specific history of trigger factors that cause DCMP except hypothyroidism; hence, he was considered to have hypothyroidism-induced DCMP. Although T4 monotherapy has become a standard treatment for hypothyroidism, his cardiac function did not improve with T4 monotherapy despite normalization of free T4 level. He was treated with T3 and T4 combination therapy, and his symptoms and cardiac function gradually improved three weeks after initiation of combination therapy, and he fully recovered after 6 months.
The effect of TH on the heart is closely related to cardiac function through cellular mechanisms. TH transporters such as Na+/K+-ATPase, Na+/Ca2+ exchanger, several voltage-gated potassium channels, monocarboxylate transporter (MCT) 8, and MCT10 have been identified in different cells and organs [12]. They are highly specific for iodothyronines, and the transport mechanism for TH appears to be facilitative diffusion [13]. However, recent data suggest that T4 is not transported into the heart [14]. In addition, TH affects cardiac myocyte-specific gene transcription mediated by TH receptors [15]. These receptor proteins are bound to TH response elements in the promoter region of specific T3-responsive genes and activate transcription of positively regulated genes in the presence of T3 [16]. Therefore, T3 genomically regulates multiple regulatory and structural myocyte genes related to cardiac contractile function [3]. Consequently, T3 is more important than T4 in a cardiac cellular mechanism. In addition to the genomic effects, TH has a nongenomic effect on cardiac myocytes by altering the functional properties of membrane ion channels and pumps [14]. T3 appears to reduce systemic vascular resistance by directly affecting vascular smooth muscle cells and through changes in the vascular endothelium, potentially involving the synthesis and secretion of nitric oxide [17]. Therefore, clinicians should consider T3 and T4 combination therapy for patients with hypothyroidism accompanied by heart disease.
In summary, we report the case of a teenage boy with hypothyroidism-induced DCMP who was successfully managed with combination therapy of T3 and T4. Moreover, as there are no guidelines for T3 combination therapy, and since this is the first report of T3 combination therapy in an adolescent with hypothyroidism-induced DCMP, our case would be a good example of T3 combination therapy in adolescents. However, further studies are needed to establish a guide for T3 combination therapy for hypothyroidism accompanying heart disease.
Notes
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contribution
Conceptualization: YC, SYJ, JMP, JS, HWC, HK, AK; Data curation: YC, EJS, AK; Formal analysis: YC, AK; Funding acquisition: YC, AK; Methodology: YC, AK; Project administration: YC, AK; Visualization: YC, AK; Writing - original draft: YC, SYJ, AK; Writing - review & editing: YC, AK
Fig.┬Ā1.
Chest x-ray on the first day after transfer. Chest x-ray showed cardiomegaly and right pleural effusion in initial chest x-ray (A) and showed normal findings at 12 months of follow-up (B).
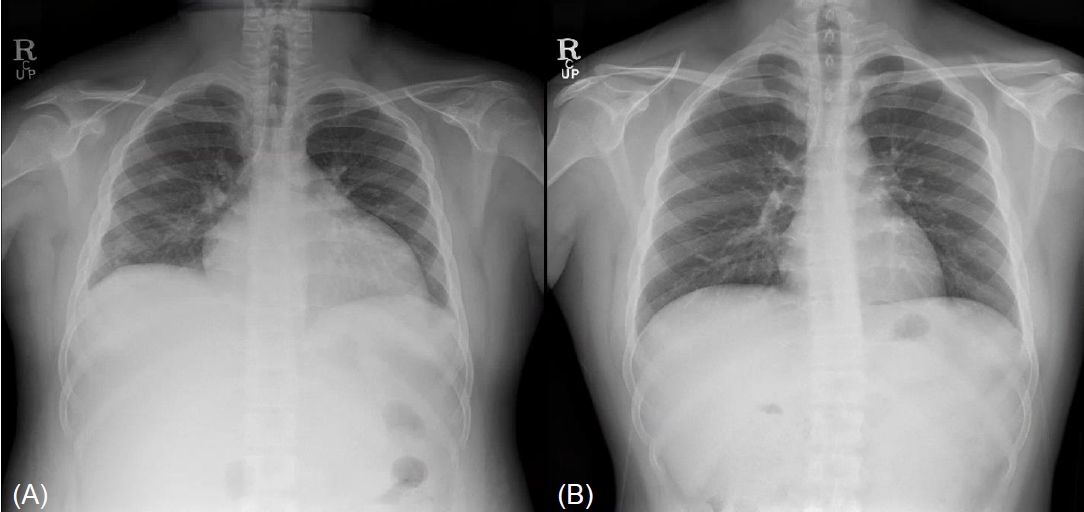
Fig.┬Ā2.
Markedly enlarged left ventricle with moderate mitral regurgitation was seen in the initial apical four-chamber view via transthoracic echocardiography (A), and decreased left ventricle to normal range without mitral regurgitation was seen in the same view at 12 months of follow-up (B).

Fig.┬Ā3.
The 12-lead electrocardiography findings. The patient had sinus tachycardia in initial electrocardiography (A) and normal sinus rhythm at 12 months of follow-up (B).

Table┬Ā1.
Summary of the laboratory findings, echocardiographic results, and medication
References
2. Mullur R, Liu YY, Brent GA. Thyroid hormone regulation of metabolism. Physiol Rev 2014;94:355ŌĆō82.



4. Cohn JN, Bristow MR, Chien KR, Colucci WS, Frazier OH, Leinwand LA, et al. Report of the National Heart, Lung, and Blood Institute Special Emphasis Panel on Heart Failure Research. Circulation 1997;95:766ŌĆō70.


5. Davis PJ, Davis FB. Acute cellular actions of thyroid hormone and myocardial function. Ann Thorac Surg 1993;56(1 Suppl):S16ŌĆō23.


6. Klein I, Danzi S. Thyroid hormone treatment to mend a broken heart. J Clin Endocrinol Metab 2008;93:1172ŌĆō4.



7. Henderson KK, Danzi S, Paul JT, Leya G, Klein I, Samarel AM. Physiological replacement of T3 improves left ventricular function in an animal model of myocardial infarction-induced congestive heart failure. Circ Heart Fail 2009;2:243ŌĆō52.


8. Brent G, Davies T, Larsen P. Hypothyroidism and thyroiditis. Kronenberg HM, Melmed S, Polonsky KS, Larsen PRet al., editors. Williams textbook of endocrinology. 11th ed. Philadelphia (PA): Elsevier. 2008;pp 406ŌĆō35.
9. Wang W, Guan H, Gerdes AM, Iervasi G, Yang Y, Tang Y-DJTJoCE, et al. Thyroid status, cardiac function, and mortality in patients with idiopathic dilated cardiomyopathy. J Clin Endocrinol Metab 2015;100:3210ŌĆō8.



10. Bozkurt B, Colvin M, Cook J, Cooper LT, Deswal A, Fonarow GC, et al. Current diagnostic and treatment strategies for specific dilated cardiomyopathies: a scientific statement from the American Heart Association. Circulation 2016;134:e579ŌĆō646.


11. Bezdah L, Slim├©ne H, Kammoun M, Haddad A, Belhani A. Hypothyroid dilated cardiomyopathy. Ann Cardiol Angeiol (Paris) 2004;53:217. ŌĆō20. French.

12. Jansen J, Friesema EC, Kester MH, Schwartz CE, Visser TJ. Genotype-phenotype relationship in patients with mutations in thyroid hormone transporter MCT8. Endocrinology 2008;149:2184ŌĆō90.




13. Visser WE, Wong WS, van Mullem AA, Friesema EC, Geyer J, Visser TJ. Study of the transport of thyroid hormone by transporters of the SLC10 family. Mol Cell Endocrinol 2010;315:138ŌĆō45.


15. Lazar MA. Thyroid hormone receptors: multiple forms, multiple possibilities. Endocr Rev 1993;14:184ŌĆō93.


-
METRICS

-
- 1 Web of Science
- 0 Crossref
- Scopus
- 2,397 View
- 142 Download






