Introduction
Vitamin D is not just for preventing rickets and osteomalacia. Recent findings, primarily in animals studies but also in a number of epidemiologic studies and a few clinical trials, indicate that adequate vitamin D levels are important for normal growth and puberty, cancer prevention, controlling hormones such as parathyroid hormone (PTH), insulin, fibroblast growth factor 23 (FGF23), and renin, and regulation of the immune response. 25-hydroxyvitamin D (25(OH)D) levels above 10 ng/mL may suffice to prevent rickets and osteomalacia, but these levels are not sufficient to provide for these more recently discovered clinical benefits. The recent report indicate that a level of 20 ng/mL was adequate for maintenance of skeletal health, but the evidence was insufficient to make recommendations for the nonskeletal actions, but determination of the optimal levels of 25(OH)D and thus amount of vitamin D supplementation required to reach these levels for the numerous actions of vitamin D will only be established with additional prospective trials. The objectives of this review are to understand the basic metabolism and mechanisms of action of vitamin D, including its pleiomorphic actions in multiple tissues. And to assess the quality of the data supporting the current recommendations for serum 25(OH)D levels, and the know where the gaps in our knowledge exist regarding optimal vitamin D levels. Last, to determine the appropriate means of achieving vitamin D sufficiency with supplementation, while appreciating some of the pitfalls regarding monitoring those levels deemed sufficient.
The background of physiology of vitamin D
The large amount of work by a number of laboratories near 10 decades demonstrated that vitamin D not only was essential for bone health but also was a hormone mediating a wide range of homeostatic functions.
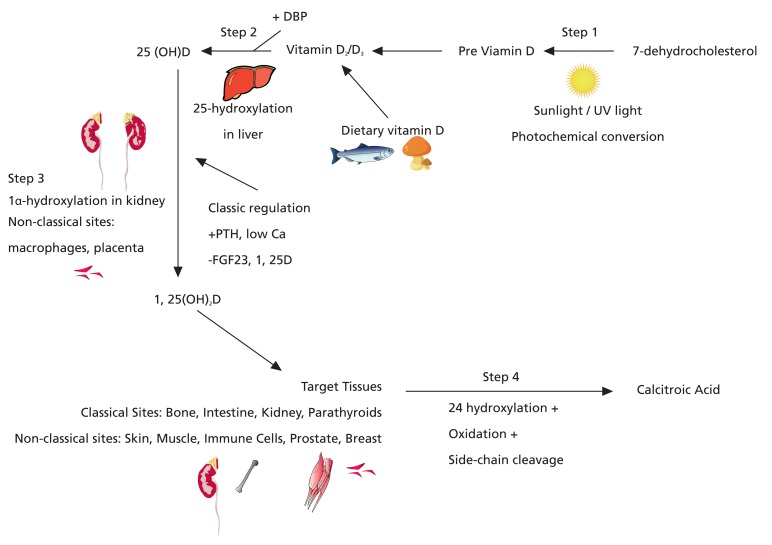
The physiology of vitamin D from its synthesis in the skin to its active form 1,25-dihydroxyvitamin D (1,25(OH)2D), was fully defined by the mid-1970s (Fig. 1)1). The cloning of the vitamin D receptor (VDR) occurred by 1987. After that, in all tissues, further basic and clinical studies uncovered subsequent actions and led to the surprisingly abundant physiological role of vitamin. Simultaneously, vitamin D as a therapeutic modality for the prevention of chronic diseases was rising. In a recent 2-year, there were more than 500 publications centered on vitamin D. In 2012, an Endocrine Society Scientific Statement was published in Endocrine Review2). The evidence that defines the effects of vitamin D on epidermal, neuromuscular, cardiovascular, metabolic, immunological, maternal/fetal, and neoplastic tissues1). In Fig. 2, the metabolic pathway of vitamin D, including the various steps in its activation and degradation, are depicted. It was assumed that exposure of the skin to ultraviolet (UV) radiation drove the conversion of 7-dehydrocholesterol to cholecalciferol (Fig. 2, step 1). The photo-production is subject to a variety of factors including amount of UV exposure (latitude, season, and use of sunscreen and clothing), ethnicity (skin pigmentation), and age3). After the photochemical conversion of 7-dehydrocholesterol to previtamin D3 and its thermal isomerization to vitamin D3, it binds to the vitamin D-binding protein (DBP), and is transported to the liver where a hydroxyl group is attached at the carbon-25 atom (i.e., C-25) to generate 25(OH)D (Fig. 2, step 2). A number of 25-hydroxylases have been reported including cytochrome P450 CYP27A1 and CYP2R14,5). CYP2R1 is probably the major enzyme required for 25-hydroxylation of vitamin D3, at least in humans6). A patient with classic rickets and low circulating levels of 25(OH) D was found to have a homozygous mutation of the CYP2R1 gene, implying that other enzymes were unable to compensate7). Much remains unknown about the 25-hydroxylase enzymes including the significance of their reported presence in skin, kidney, and intestine8). In the absence of severe hepatic dysfunction, 25-hydroxylation of vitamin D is not usually rate limiting. However, in mild to moderate liver impairment, the associated fat malabsorption can cause vitamin D deficiency. In contrast, 1╬▒-hydroxylation is the major rate-limiting step in synthesis of 1,25(OH)2D (Fig. 2, step 3). Synthesis of 1,25(OH)2D is tightly regulated9) and is mediated by the enzyme 1╬▒-hydroxylase. Factors regulating 1╬▒-hydroxylase in kidney are shown. Due to its sequence similarity to CYP27A1, the gene encoding 1╬▒-hydroxylase was called CYP27B110). Its role was demonstrated in 1998 by the development of rickets and reduced circulating 1,25(OH)2D levels in four patients with gene mutations for this enzyme11). Cyp27B1-null mice also develop rickets with reduced levels of circulating 1,25(OH)2D12). CYP27B1 mRNA is expressed in a number of vitamin D target tissues including kidney, skin, intestine, macrophages, and bone. Although its expression is relatively high in skin, the kidney is thought to be primarily responsible for circulating levels of 1,25(OH)2D8). This is supported by 1,25(OH)2D deficiency in people with renal failure13). However, this has not been conclusively proven with renal-specific CYP27B1 deletion. The presence of CYP27B1 in other cell types, especially macrophages, is demonstrated by the fact that people with granulomatous diseases can have elevated 1,25(OH)2D levels14).
Entry of 25(OH)D into the proximal renal tubular cells requires receptor-mediated uptake of DBP plus 25OHD at the brush border, degradation of DBP by legumain, and endocytic internalization and translocation of 25(OH)D to mitochondria15). Megalin, a member of the low-density lipoprotein receptor family, is required for reabsorption of filtered DBP-bound 25OHD16). It is in the mitochondria that 1╬▒-hydroxylation of 25(OH)D into its biologically active form, 1,25(OH)2D, occurs8). A number of factors contribute to the tight regulation of 1╬▒-hydroxylase enzyme expression and activity in the kidney (Fig. 2). These include calcium, PTH, calcitonin, growth hormone (GH), insulin-like growth factor-I (IGF-I), and FGF23. In addition, 1,25(OH)2D negatively regulates its own synthesis by suppressing 1╬▒-hydroxylase expression in kidney and bone17). There is also evidence to suggest that estrogen, progesterone, and prolactin may regulate 1╬▒-hydroxylase activity18,19). In macrophages, regulation of CYP27B1 is primarily cytokine mediated20). The final important enzyme in the vitamin D endocrine system is 24-hydroxylase (CYP24A1). Found in nearly all cells and highly expressed by the kidney, CYP24A1 limits the amount of 1,25(OH)2D in target tissues by converting 1,25(OH)2D to inactive metabolites, including 1,24,25(OH)3D and calcitroic acid and by converting 25D to 24,25(OH)2D (Fig. 2, step 4). In addition to 24-hydroxylation, this multicatalytic enzyme is able to catalyze side-chain hydroxylations at the C23 and C26 positions21). Recently, mutations in CYP24A1 were reported in six children with infantile hypercalcemia, thereby providing conclusive evidence of the importance of this enzyme in the in vivo regulation of vitamin D metabolism22).
VDR
The VDR represents the final common pathway through which vitamin D works on target tissues. So, we need to know the physiology of the VDR (Fig. 3)2). The family of molecules known collectively as vitamin D are not true vitamins, which are defined as essential substances obtained exclusively from the diet. Vitamin D is a steroid hormone, and the active metabolite, 1,25(OH)2D, is the ligand for a transcriptional factor. Intracellular receptor called the "vitamin D receptor". The VDR is widely distributed across many tissues. This widespread distribution underlies the potential myriad of physiologic actions for vitamin D. 1,25(OH)2D are mediated by the VDR acting primarily by regulating the expression of genes whose promoters contain specific DNA sequences known as vitamin D response elements (VDRE). The VDR works in partnership with other transcriptional factors, the best-studied of which is the retinoid X receptor (RXR), and a number of coactivators and corepressors that provide context, tissue, and target gene specificity (Fig. 4). However, some actions of 1,25(OH)2D are more immediate and may be mediated by a membrane-bound VDR that has been less well characterized than the nuclear VDR. Our understanding of the mechanism by which VDR regulates gene expression has increased enormously over the past few years.
VDR structure
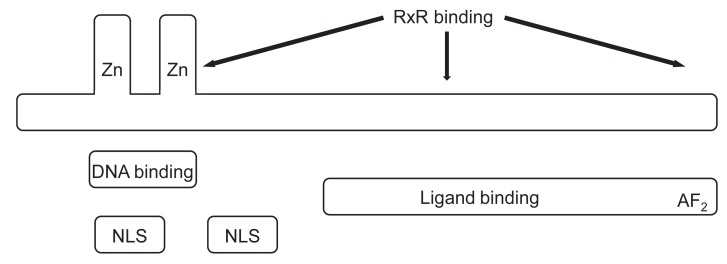
VDR is a member of a large family proteins (>150 members) that includes receptors for the steroid hormones, T4, the vitamin A family of metabolites (retinoid), fatty acids, and eicosanoids (Fig. 3). A large number of family members have no known ligands and are called orphan receptors23,24). VDR is widely, although not universally, distributed throughout different tissues of the body25,26). The human VDR has two potential start sites. A common polymorphism (Fok I) alters the first ATG start site to ACG. This polymorphism has been correlated with reduced bone mineral density (BMD) in some studies, whereas other genome-wide association studies have not found a strong signal for polymorphisms in the VDR gene and bone mass or fractures27). The most conserved domain in VDR from different species and among the nuclear hormone receptors in general is the DNA-binding domain. This domain consists of two zinc finger confers. The proximal (N-terminal) zinc finger confers specificity for DNA binding to the VDRE, whereas the second zinc finger and the region following provide at least one of the sites for heterodimerization of the VDR to the RXR. The second half of the molecule if the ligand-binding domain (LBD), the region responsible for binding 1,25(OH)2D, but also contains regions necessary for heterodimerization to RXR.
At the C-terminal end is the major activation domain, AF2, which is critical for the binding to coactivators such as those in the steroid receptor coactivator and VDR-interacting protein (also known as Mediator) families28). In mutation studies of the homologous thyroid receptor, corepressors were found to bind in overlapping regions with coactivators in helices 3 and 5, a region blocked by helix 12 (the terminal portion of the AF2 domain) in the presence of ligand29). Deletion of helix 12 promoted corepressor binding while preventing that of coactivators29). The LBD for VDR has been crystallized, and its structure solved30). It shows a high degree of structural homology to other nuclear hormone receptors. It comprises 12 helices joined primarily by ╬▓-sheets. The ligand 1,25(OH)2D is buried deep in the ligand-binding pocket and covered with helix 12 (the terminal portion of the AF2 domain). Assuming analogy with the unliganded LBD of RXR╬▒ and the ligand-bound LBD of RXR╬│, the binding of 1,25(OH)2D to the VDR triggers a substantial movement to helix 12 from and open position to a close position, covering the ligand-binding pocket and putting helix 12 in position with critical residues from helices 3, 4, and 5 to bind coactivators31).
Nongenomic actions of vitamin D
A variety of hormones that serves as ligands for nuclear hormone receptors also exert biologic effects through membrane receptors or the VDR situated outside of the nucleus. Examples include estrogen, progesterone, testosterone, corticosteroids, and thyroid hormone32). 1,25(OH)2D also have rapid effects that be mediated by a distinct receptor which is on the membrane receptor. 1,25(OH)2D also has been shown to regulate calcium and chloride channel activity, protein kinase C activation and distribution, and phospholipase C activity in a number of cells including osteoblasts, liver, muscle, and intestine33). A putative membrane receptor for 1,25(OH)2D-i.e., 1,25(OH)2D membrane-associated rapid response steroid-binding protein (1,25D-MARRSBP), also known as endoplasmic reticulum stress protein 57-has purified from the intestine, cloned, and sequenced, and blocking antibodies have been prepared that block the rapid actions of 1,25(OH)2D34). However, these rapid actions of 1,25(OH)2D appear to require the VDR (ineffective in VDR null mice), which suggests that 1,25D-MARRSBP and VDR cooperate in mediating these acute actions of 1,25(OH)2D but without the need for new protein synthesis. In the latter case, analogs of 1,25(OH)2D that do not support genomic actions of 1,25(OH)2D do support these nongenomic actions, which suggests that the membrane VDR may have a different three-dimensional structure with a different binding pocket for its activating ligands.
Growth and development
Maternal vitamin D status regulates skeletal growth and development during fetal life35). These effects partly persist in early childhood, as has been suggested in a longer prospective study36,37). In these reports, tibia cross sectional area, bone mineral content or BMD remained larger in infants whose mothers had better vitamin D status during pregnancy. Besides genetic background, bone size is affected by various hormones and it has been shown that GH-IGF-I axis is responsible for bone size38) and periosteal expansion35,39). Leptin may favor stem cell differentiation towards osteoblasts rather than adipocytes40) in infancy. Furthermore, vitamin D stimulates steoblastogenesis in human mesenchymal stem cells and production of IGF-I in osteoblasts41). In infants with rickets vitamin D supplementation increased serum IGF-I and accelerates linear growth42).
Obesity
Obesity is a strongly correlated with vitamin D deficiency. The nature of this association is not yet clear, it may be that vitamin D deficiency contributes to fat accumulation by suppression of certain key enzymes in adipose tissue differentiation (e.g., lipoprotein lipase, fatty acid synthase)43). Vitamin D deficiency may also be a consequence of obesity. Vitamin D may be "sequestered" in fat tissue44). This term has used as a possible explanation for why obese individuals had a smaller vitamin D increase than leaner individuals after UV exposure. The term "sequestration" means that vitamin D is tightly bound within fat tissue, but there is no evidence that this is the case44). In contrast, lower vitamin D levels among larger individuals are better explained by total body weight than adipose tissue weight. This suggest that "dilution" may be a better word than "sequestration" to explain the lower vitamin D values of obese people. The study of 242 girls of the Bogota School Children Cohort, Colombia, their vitamin D levels were adjusted for baseline body mass index (BMI)-for-age z score, a measure that reflects larger weight for a given height and age, not absolute weight or body pool size. This finding could reflect residual confounding by body size45). Larger girls could have lower 25(OH)D concentrations due to increased tissue dilution of vitamin D over their larger pool size and reach menarche earlier because their greater size reflects greater maturity and/or adiposity45).
Age at menarche
A geographic north-south gradient in age at menarche was described46); girls who live at higher latitudes appear to have an earlier menarche than girls who live closer to the equator47). This pattern might be explained by differences in temperature, light-darkness rhythms, and socioeconomic conditions, it also corresponds with a geographic gradient on sun exposure that coincided with vitamin D status48). Vitamin D deficiency is associated with the development of adiposity in children49), and childhood obesity could be a risk factor for early puberty50); vitamin D might play a role in the timing of puberty
To investigate the relation between vitamin D and early puberty, by Villamor et al.45) used a subset of 242 girls (mean age, 8.8┬▒1.6 years) from the Bogota School Children Cohort, which was comprised of 3,202 students (aged 5-12 years) who were representative of the population enrolled in the public school system in Bogota, Columbia (latitude 4┬░43'N). The cohort was followed for 2.5 years to determine associations between nutrition and health50). The subset included girls in whom serum 25(OH)D had been measured at baseline in February 2006. Baseline vitamin D status was categorized as <50 nmol/L (deficient, <20 ng/mL), 50 and <75 nmol/L, and 75 nmol/L (sufficient, >30 ng/mL). Follow-up assessments were conducted, twice in 2006 and once yearly in 2007 and 2008, to determine menarcheal status. Baseline 25(OH)D deficiency was correlated with a decreased age of menarche. This relationship was partially attenuated after adjustments for age at baseline and BMI-for-age a score. Except this report, no prior studies have directly examined the association of age at menarche with vitamin D level. And a gradient effect has not been observed consistently worldwide51). In Europe, the more northern latitudes are associated with later menarche, though this could be due to the stronger influence of genetic factors over environmental factors52). Twin studies indicate that as much as 68% of the variance in age at menarche may be generally determined53), and polymorphisms in the VDR gene may be involved. VDR knockout mice show both impaired ovarian development and alterations in vitamin D metabolite levels compared to wild-type mice54). In humans, the A allele of the VDR ApaI locus and the t allele of the VDR Taq 1 locus, which are linked, may be associated with earlier menarche55).
Musculoskeletal development
The clinical and radiographic signs of rickets due to severe vitamin D deficiency have been well described56). Bone mineral accretion may be compromised by vitamin D insufficiency (VDI)57), and vitamin D influences prenatal and postnatal linear growth58). Vitamin D may also affect musculoskeletal development, as high-dose supplementation (50 ┬Ąg/day) of schoolchildren aged 10-17 year-old in Beirut was found to increase lean tissue mass compared with children receiving lower-dose vitamin D (5 ┬Ąg/day) after 1 year59).
Regulation of immune responses and inflammatory diseases
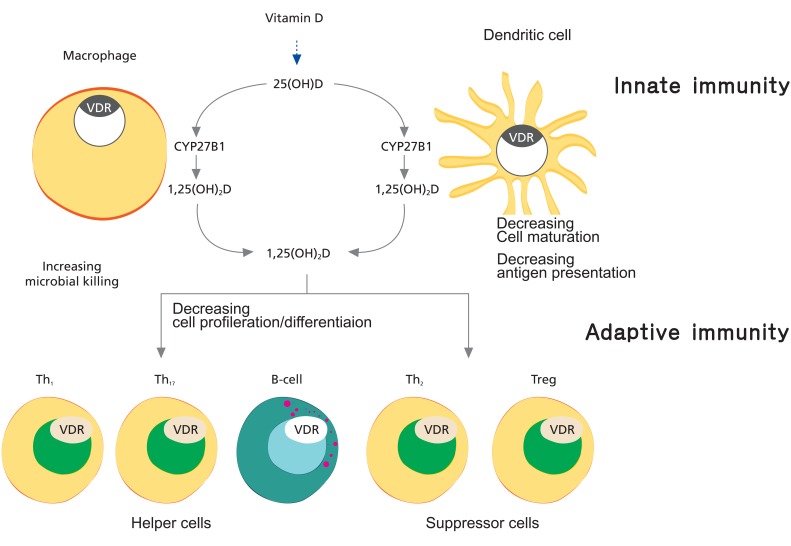
The association of VDI with immune dysregulation is compelling, because 1,25(OH)2D potentially modulates the activity of leukocytes and cytokine production60). Many of the actions of 1,25(OH)2D are anti-inflammatory and antiproliferative, including the downregulation of proinflammatory tumor necrosis factor-╬▒ synthesis61) and nonspecific reduction of T cell responsiveness62). The clinical implications for children and adolescents are unclear, yet there is evidence that VDI in adults predisposes to obesity-related conditions such as insulin resistance63), possibly related to the role of vitamin D in modulating chronic inflammatory responses64). In vitro observations suggest that 1,25(OH)2D suppresses T helper lymphocyte type 1 (Th1) cytokine production in favour of Th2 cytokines65) (Fig. 5). Administration of 1,25(OH)2D prevented and arrested66) the Th1-mediated immune response implicated in diabetes pathogenesis in nonobese diabetic mice. Epidemiologic studies in Finland have found that vitamin D supplementation during infancy was associated with a reduced risk of type I diabetes67), but an increased risk of atopic disease (Th2 mediated) in adulthood68), suggesting that the effect of vitamin D on T cell development during infancy may have longterm clinical implications. Clinical trials to clarify these associations have not yet been reported.
Vitamin D status and the risk of lower respiratory tract infection
Rickets commonly co-occurs with pneumonia69), yet few studies have explored the possibility of a direct causal role of VDI in susceptibility or severity of childhood acute lower respiratory infection (ALRI). Accumulating evidence points towards antimicrobial properties of vitamin D; for example, the expression of an innate antimicrobial peptide, cathelicidin antimicrobial peptide is dependent on 1,25(OH)2D activity70). A case-control study conducted in India showed that vitamin D deficiency was a significant independent risk factor for severe ALRI in children under 5 years of age71). Among children aged 1-24 months in Edmonton, they did not find vitamin D deficiency alone to be associated with the risk of ALRI (mostly viral bronchiolitis) in a relatively vitamin D repleted group, but the combination of a hypofunctional polymorphic variant of the VDR and 25(OH)D concentration <80 nmol/L was significantly associated with the risk of ALRI72). In Northern Aboriginal communities at high risk of VDI, where an extraordinarily high incidence of severe infantile bronchiolitis requiring hospitalization has been reported73), it remains possible that moderate to severe VDI contributes to immune dysregulation, which predisposes to over-exuberant host responses to viral pathogens.
Dietary vitamin D requirements of the child and adolescent
Dietary intake of vitamin D is necessary if endogenous synthesis is limited (e.g., northern latitude, clothing habits, or sedentar y lifestyles that limit sunlight exposure). However, adequate vitamin D nutriture often depends on the consumption of commercially fortified products because vitamin D naturally occurs in foods that are scarce or infrequently consumed. Vitamin D2 (ergocalciferol) is found primarily in UV-exposed fungi and some vitamin D supplements; vitamin D3 (cholecalciferol) is found in some animal tissues (i.e., liver, sea fish oils, and egg yolks), is used in most fortified products (e.g., milk) and is the form produced endogenously in human epidermis.
Hollis and Wagner74) have convincingly contended that a 70 kg adult and a 1 kg premature infant have the same absolute dietary vitamin D requirements. In fact, doses of at least 50 ┬Ąg/day (20,00 IU/day) may be required to maintain vitamin D sufficiency in adults75). A daily dose of 100 ┬Ąg/day (4,000 IU/day) for 2-5 months was shown to be safe in adult.
The level of 25(OH)D in the blood is the best indicator of vitamin D status. There is consensus that levels below 25 nmol/L (10 ng/mL) qualify as 'deficient76), but beyond this is currently no standard definition of 'optimal' 25(OH)D levels77). There is also lack of standardization of methods used to measure 25(OH) D status, with different tests producing very different results78). Some scientists suggest that levels above 50 ng/mL (20 ng/mL) are 'sufficient", while 70-80 nmol/L (28-32 ng/mL) is 'optimal'76). However, raising the definition of 'deficiency' or 'sufficiency' is currently inappropriate since no results from randomized trials suggest that maintaining such levels of 25(OH)D prevents chronic diseases. It is also unclear whether these levels are practical for all individuals. Given that various studies have found that that 25(OH)D levels plateau at around 70-80 nmol/L, with wide variation across individuals79). For example, a Hawaiian study found that half of healthy, young surfurs had levels below 75 nmol/L despite extensive unprotected outdoor exposure and tanned complexions79). Most recommendations are daily 10 microgram vitamin D supplement for those at risk of vitamin D deficiency, including all pregnant and breastfeeding women, older people and those at risk of inadequate sun exposure. A daily vitamin D supplement of 7 micrograms is also recommended for all children aged 6 months to 5 years80). The importance of maintaining adequate vitamin during pregnancy and breastfeeding suggests that women may choose to take up to 10 g of vitamin D a day during these periods81).
Vitamin D requirements for infant
25(OH)D concentration of between 40 and 50 nmol/L should be considered for vitamin D sufficiency. For toddlers and children older than 1 year of age, estimated average requirement is 400 IU/day (10 ┬Ąg/day). Dietary Allowance of 600 IU/day (15 ┬Ąg/day) are proposed for infants less than 1 year. Breast-milk contains limited amounts of vitamin D, so lactating mothers with high levels of vitamin D (2,000-6,400 IU/day) may maintain vitamin D sufficiency. Breastfed infants should be supplemented with 400 IU/d of vitamin D. In United Kingdom, infants up to 6 months of 320 IU/day (8 ┬Ąg/day) for infants and children aged 6-36 months of 280 IU/day (7 ┬Ąg/day).
Conclusions
Vitamin D is not only essential for bone health and growth, but also hormone mediating nonskeletal effects across a wide range of homeostatic functions. Pharmacokinetic studies and epidemiologic research that incorporates clinical and functional outcomes are needed to clarify the role of vitamin D in growth and development and the specific dietary vitamin D requirements among Korean children and adolescents.