 |
 |
- Search
| Ann Pediatr Endocrinol Metab > Volume 29(1); 2024 > Article |
|
Abstract
Purpose
Methods
Results
Notes
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Fig.┬Ā1.
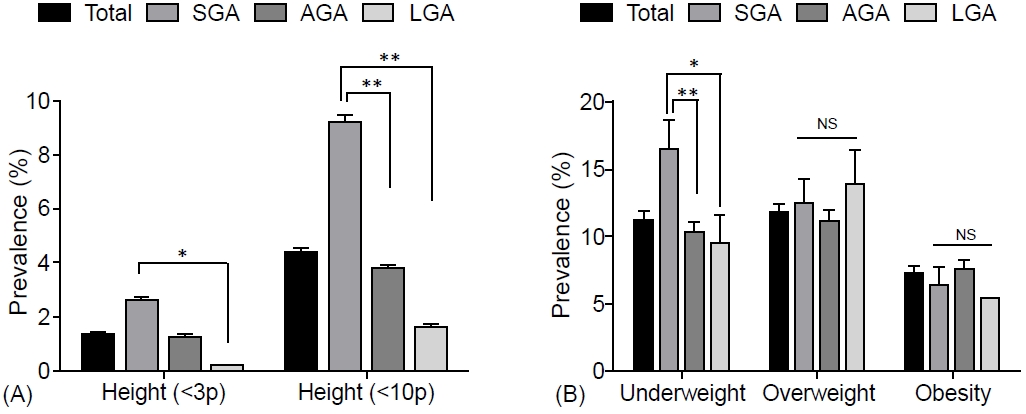
Fig.┬Ā2.

Table┬Ā1.
| Variable | SGA (N=272) | AGA (N=1,286) | LGA (N=190) | P-value* |
|---|---|---|---|---|
| Age (yr) | 13.8┬▒2.6 | 13.6┬▒2.5 | 13.8┬▒2.5 | 0.333 |
| Male sex | 139 (50.9) | 700 (54.3) | 92 (48.4) | 0.230 |
| Height (cm) | 157.8┬▒12.0a) | 159.6┬▒11.7a) | 162.4┬▒10.4b) | <0.001 |
| Weight (kg) | 50.5┬▒13.7a) | 52.7┬▒14.1b) | 55.8┬▒13.9c) | <0.001 |
| BMI (kg/m2) | 20.0┬▒3.7a) | 20.4┬▒3.7a,b) | 20.9┬▒3.5b) | 0.024 |
| WC (cm) | 67.4┬▒10.0a) | 68.7┬▒10.1a,b) | 70.0┬▒9.1b) | 0.020 |
| SBP (mmHg) | 105.6┬▒9.8 | 105.6┬▒11.0 | 105.9┬▒9.4 | 0.912 |
| DBP (mmHg) | 64.9┬▒9.9 | 65.7┬▒9.1 | 66.1┬▒9.0 | 0.316 |
| TG (mg/dL) | 82.1┬▒40.2 | 82.8┬▒50.7 | 77.7┬▒41.0 | 0.435 |
| TC (mg/dL) | 155.8┬▒24.7 | 158.6┬▒26.9 | 159.8┬▒25.0 | 0.236 |
| HDL-C (mg/dL) | 53.6┬▒9.7 | 54.3┬▒10.6 | 55.7┬▒11.2 | 0.138 |
| ALT (IU/L) | 13.9┬▒10.2 | 15.4┬▒15.8 | 14.0┬▒10.1 | 0.248 |
| FPG (mg/dL) | 88.9┬▒6.7 | 89.1┬▒9.6 | 88.9┬▒5.3 | 0.925 |
| Insulin (╬╝IU/mL) | 13.4┬▒5.6 | 14.0┬▒6.8 | 13.4┬▒5.6 | 0.546 |
| HOMA-IR | 3.0┬▒1.4 | 3.1┬▒1.5 | 3.0┬▒1.3 | 0.701 |
| Birth history | ||||
| ŌĆāBirth weight (g) | 2644.3┬▒265.3a) | 3278.3┬▒318.7b) | 3927.1┬▒534.8c) | <0.001 |
| ŌĆāŌĆāMacrosomia | 0 (0)a | 6 (5.4)a) | 105 (94.6)b) | <0.001 |
| ŌĆāŌĆāLBW | 40 (14.7)a) | 16 (1.2)b) | 6 (3.2)b) | <0.001 |
| ŌĆāŌĆāVLBW | 2 (0.7) | 2 (0.2) | 1 (0.5) | 0.215 |
| ŌĆāGA (wk) | 39.5┬▒1.3a) | 39.6┬▒1.4a) | 39.0┬▒2.9b) | <0.001 |
| ŌĆāŌĆāPremature | 11 (4.0)a) | 36 (2.8)a) | 18 (9.5)b) | <0.001 |
| ŌĆāŌĆāPostmature | 3 (1.1) | 43 (3.4) | 7 (3.7) | 0.125 |
| ŌĆāMaternal age (yr) | 27.7┬▒4.1 | 28.2┬▒4.1 | 28.4┬▒4.0 | 0.078 |
| ŌĆāC/Sec | 95 (34.8) | 472 (36.6) | 77 (40.5) | 0.445 |
| Household income (1ŌĆō4)ŌĆĀ | 2.8┬▒1.0 | 2.9┬▒1.0 | 2.9┬▒1.0 | 0.218 |
| Physical activity, none | 140 (51.3) | 684 (53.2) | 94 (49.7) | 0.602 |
| Current smoking | 12 (3.7) | 64 (5.0) | 16 (8.4) | 0.108 |
| Current alcoholŌĆĪ | 50 (24.2) | 184 (15.3) | 38 (25.7) | 0.211 |
| Calorie intake (kcal/day) | 2156.3┬▒856.0 | 2176.1┬▒860.2 | 2217.3┬▒860.3 | 0.772 |
| Food intake | ||||
| ŌĆāCarbohydrate (g/day) | 331.9┬▒128.0 | 336.6┬▒124.0 | 340.9┬▒129.8 | 0.785 |
| ŌĆāProtein (g/day) | 77.3┬▒38.0 | 78.4┬▒42.2 | 79.0┬▒39.5 | 0.905 |
| ŌĆāFat (g/day) | 58.2┬▒37.9 | 57.3┬▒36.5 | 59.0┬▒38.5 | 0.815 |
Values are presented as mean┬▒standard deviation or number (%).
SGA, small for gestational age; AGA, appropriate for gestational age; LGA, large for gestational age; BMI, body mass index; WC, weight circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; TG, triglyceride; TC, total cholesterol; HDL-C, high-density lipoprotein-cholesterol; ALT, alanine transaminase; FPG, fasting plasma glucose; HOMA-IR, homeostasis model of assessmentŌĆōinsulin resistance; LBW, low birth weight; VLBW, very low birth weight; GA, gestational age; C/sec, cesarean section.
Table┬Ā2.
The odds ratios of short stature and underweight in Korean children and adolescents according to birth weight at gestational age from multiple logistic regression analysis.
Model 1, nonadjusted; model 2, adjusted for age and sex; model 3, adjusted for age, sex, low birth weight, and prematurity; OR, odds ratio; CI, confident interval; SGA, small for gestational age; AGA, appropriate for gestational age; LGA, large for gestational age; BMI, body mass index.
Table┬Ā3.
| Variable |
Male (N=642) |
Female (N=550) |
||||||
|---|---|---|---|---|---|---|---|---|
| SGA (n=102) | AGA (n=473) | LGA (n=67) | P-value* | SGA (n=87) | AGA (n =401) | LGA (n=62) | P-value* | |
| Anthropometric parameter | ||||||||
| ŌĆāAge (yr) | 13.6┬▒2.5 | 13.6┬▒2.5 | 13.6┬▒2.5 | 0.968 | 13.7┬▒2.6 | 13.5┬▒2.5 | 13.6┬▒2.6 | 0.816 |
| ŌĆāHeight (cm) | 161.4┬▒14.2 | 161.9┬▒13.1 | 164.1┬▒13.3 | 0.406 | 154.5┬▒7.3a) | 156.4┬▒8.8a) | 159.5┬▒7.0b) | 0.002 |
| ŌĆāWeight (kg) | 54.0┬▒15.2 | 55.2┬▒15.6 | 56.1┬▒13.6 | 0.678 | 46.6┬▒10.1a) | 49.7┬▒11.5a,b) | 52.2┬▒11.4b) | 0.010 |
| ŌĆāBMI (kg/m2) | 20.4┬▒3.8 | 20.7┬▒3.9 | 20.5┬▒3.0 | 0.758 | 19.4┬▒3.3 | 20.1┬▒3.5 | 20.4┬▒3.7 | 0.144 |
| Body composition | ||||||||
| ŌĆāLean mass (kg) | 40.6┬▒10.9 | 41.1┬▒11.1 | 41.9┬▒10.7 | 0.749 | 31.6┬▒5.3 | 33.2┬▒6.3 | 34.5┬▒5.2 | 0.014 |
| ŌĆāLean mass ratioŌĆĀ (%) | 75.6┬▒8.1 | 75.2┬▒8.3 | 74.7┬▒6.4 | 0.757 | 68.8┬▒5.9 | 67.6┬▒5.5 | 67.2┬▒7.0 | 0.185 |
| ŌĆāLMI (kg/m2) | 15.2┬▒2.2 | 15.4┬▒2.2 | 15.3┬▒2.1 | 0.867 | 13.2┬▒1.5 | 13.5┬▒1.6 | 13.5┬▒1.4 | 0.280 |
| ŌĆāFat mass (kg) | 13.1┬▒6.7 | 13.6┬▒7.1 | 13.7┬▒5.0 | 0.783 | 14.6┬▒5.4a) | 16.1┬▒5.9a,b) | 17.4┬▒7.1b) | 0.016 |
| ŌĆāFat mass ratioŌĆĪ (%) | 23.7┬▒8.4 | 24.1┬▒8.4 | 24.5┬▒6.7 | 0.832 | 30.4┬▒5.8 | 31.6┬▒5.5 | 32.4┬▒7.1 | 0.098 |
| ŌĆāFMI (kg/m2) | 5.0┬▒2.4 | 5.2┬▒2.6 | 5.1┬▒1.9 | 0.836 | 6.0┬▒2.1 | 6.5┬▒2.1 | 6.8┬▒2.6 | 0.095 |
| ŌĆāTruncal fat (kg) | 5.6┬▒3.6 | 5.8┬▒3.7 | 5.7┬▒2.6 | 0.830 | 6.0┬▒2.7a) | 6.8┬▒3.3a,b) | 7.4┬▒3.7b) | 0.036 |
| ŌĆāTruncal fat ratio┬¦ (%) | 40.9┬▒5.4 | 41.0┬▒5.5 | 40.3┬▒4.9 | 0.603 | 40.2┬▒4.6 | 41.0┬▒5.0 | 41.1┬▒5.2 | 0.365 |
| ŌĆāBMCWB (kg) | 1.8┬▒0.5 | 1.9┬▒0.6 | 1.9┬▒0.6 | 0.672 | 1.6┬▒0.4 | 1.7┬▒0.4 | 1.8┬▒0.3 | 0.076 |
| Bone mineral density | ||||||||
| ŌĆāLS z-score | 0.01┬▒1.11 | -0.56┬▒1.02 | 0.01┬▒0.95 | 0.778 | -0.22┬▒1.02 | -0.01┬▒1.05 | 0.09┬▒0.88 | 0.177 |
| ŌĆāFN z-score | -0.13┬▒0.96 | -0.05┬▒1.04 | -0.01┬▒0.84 | 0.722 | -0.20┬▒0.96 | -0.02┬▒1.02 | 0.07┬▒0.92 | 0.226 |
| ŌĆāWB z-score | -0.10┬▒0.98 | -0.01┬▒0.94 | 0.02┬▒0.83 | 0.612 | -0.18┬▒0.93 | 0.00┬▒0.93 | 0.08┬▒0.77 | 0.162 |
Values are presented as mean┬▒standard deviation.
Comparison of body composition and bone density in Korean children and adolescents according to birth weight at gestational age SGA, small for gestational age; AGA, appropriate for gestational age; LGA, large for gestational age; BMI, body mass index; LMI, lean body mass index; FMI, fat mass index, BMCWB, whole-body bone mass; LS, total lumbar spine; FN femur neck; WB, whole-body bone.
References
- TOOLS
-
METRICS

-
- 1 Crossref
- Scopus
- 1,646 View
- 97 Download
- Related articles in APEM






