The association between idiopathic scoliosis and growth hormone treatment in short children
Article information
Abstract
Purpose
Idiopathic scoliosis is the most common form of scoliosis, and the risk of onset and progression has been found to correlate with growth spurts. Therefore, treatment with recombinant human growth hormone (GH) treatment in short children may initiate and/or aggravate scoliosis. The aim of this study was to investigate the relationship between idiopathic scoliosis and GH treatment in short children.
Methods
The medical records of 113 subjects seen at the participating institution between January 2010 and December 2020 and who were diagnosed with GH deficiency and small for gestational age, had idiopathic short stature, and were treated with GH for at least one year were reviewed. Scoliosis was defined as a Cobb angle greater than 10 degrees as assessed using a spine x-ray. Clinical data and laboratory findings before and 12 months after GH treatment were compared.
Results
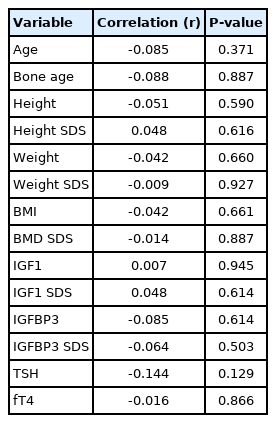
There was significant increase in height, height-standard deviation score, insulin-like growth factor 1, and insulin-like growth factor binding protein 3 (P<0.001) with GH treatment. However, there were no significant differences in the average Cobb angle (6.2°±3.3° vs. 6.1°±3.5°, P=0.842) and the prevalence of scoliosis (9.7% vs. 13.3%, P=0.481) before and after one year of GH treatment. A comparative analysis of both initial Cobb angle and change in Cobb angle during GH treatment showed no relationship with other factors.
Conclusions
Although GH treatment in short children increased height and growth velocity, it was not associated with development or aggravation of idiopathic scoliosis.
Highlights
· Idiopathic scoliosis is the most common form of scoliosis, and has been found to correlate with growth spurts.
· Recombinant human growth hormone (GH) treatment in short children promotes linear growth in short children.
· There was no association with development or aggravation of idiopathic scoliosis and GH treatment.
Introduction
Scoliosis affects 2%–5.2% of children and adolescents, and its causes vary, including genetic, environmental, hormonal, metabolic, biochemical, neurological, or idiopathic [1-4]. Idiopathic scoliosis is the most common form. Patients with idiopathic scoliosis usually develop clinical symptoms as they age, especially during puberty and growth spurts [1]. The risks of initiation and progression of idiopathic scoliosis were significantly correlated with the duration of linear rapid skeletal growth and, in particular, with peak spine growth velocity [5,6].
Patients with mild spinal curve are usually asymptomatic or may not experience clinically significant symptoms, though they are likely to have impaired pulmonary function, back pain, adverse psychologic effects, and low quality of life in the long term [1,7].
In recent decades, growth hormone (GH) treatment has been approved and used for GH deficiency (GHD), small for gestational age (SGA) without catch-up growth, idiopathic short stature (ISS), Turner Syndrome, and Prader-Willi Syndrome (PWS) [1,8,9]. GH treatment improves linear growth and can restore body composition, promote linear growth by increasing growth velocity, and improve quality of life [10-12].
Idiopathic scoliosis is one of the major concerns in GH treatment as the two might be associated. Several studies have demonstrated the impact of GH treatment on the progression of scoliosis [13,14]. However, a recent study on GH treatment and scoliosis reported that the incidence of scoliosis in children treated with GH was about 4%, similar to that of idiopathic scoliosis in the general population [15].
The aim of this study was to investigate the relationship between idiopathic scoliosis and GH treatment in short children.
Materials and methods
1. Subjects
This retrospective study reviewed the medical records of children and adolescents who were treated with GH and who attended regular follow-up visits at the pediatric endocrinology clinic at Korea University Ansan Hospital. Among the 113 subjects, 26 were diagnosed with GHD, 9 were diagnosed with SGA without catch-up growth, and 78 were diagnosed with ISS. All patients were treated with GH for at least one year between January 2010 and December 2020.
2. Definition
GHD was described as the presence of short stature with peak GH level less than 10 ng/mL in 2 provocation tests (arginine provocation and insulin-induced hypoglycemia) and delayed bone age (BA) compared to chronological age (CA) in children older than 4 years [16,17]. SGA was defined as below the third percentile of gestational age by weight without catch-up growth by the age of 4 years.
ISS was defined as height less than the third percentile in the age- and sex-matched general population after exclusion of other diseases [18]. Provocation tests were performed in all but 3 subjects.
3. Clinical data
Patients' CA, height, weight, and body mass index (BMI) were reviewed during each visit. The standard deviation scores (SDSs) for height, weight, and BMI were also calculated [19].
Laboratory findings, including levels of serum insulin-like growth factor 1 (IGF1), insulin-like growth factor binding protein 3 (IGFBP3), thyroid stimulating hormone (TSH), and free thyroxine (fT4), were examined in all subjects at baseline and after 12 months of GH treatment. Serum GH, IGF1, IGFBP3, TSH, and fT4 levels were measured using radioimmunoassay methods (Gamma Pro, Seyeong NDS Co., Seoul, Korea). The SDSs for IGF1 and IGFBP3 were the corresponding reference values from 2012 data for Korean children and adolescents [20].
Radiological assessments included BA on wrist x-rays and the Cobb angle on whole spine x-ray [21]. A single pediatric endocrinologist used the Greulich and Pyle method to assess BA on wrist x-ray images. Scoliosis was defined as a Cobb angle greater than 10 degrees [22]. To estimate the Cobb angle, the anteriorposterior (AP) view of spine x-ray images in the standing position was assessed using the angle between intersecting lines drawn perpendicular to the top of the top vertebrae and the bottom of the bottom vertebrae [23]. To reduce errors in measurements, the Cobb angle was measured 3 times in each patient, and the average value was calculated by one pediatric doctor. The follow-up BA and Cobb angle were measured after 12 months of GH treatment.
4. Statistical analysis
Continuous variables are presented as mean±standard deviation or standard error of the mean, and categorical variables were presented as number (%). The analysis of variance test was used to compare the baseline characteristics of the 3 groups, and Bonferroni test was used for multiple comparisons. The chi-square test was used for categorical variables in analysis of the 3 groups (GHD, SGA, and ISS). Anthropometric, laboratory, and radiological parameters before and after GH treatment were compared using paired t-tests. Pearson correlation test was used to investigate the relationships between the Cobb angle and multiple parameters. The McNemar test was used to compare the prevalence of idiopathic scoliosis before and after GH treatment. A P-value <0.05 was considered statistically significant. Data analysis was performed using IBM SPSS Statistics ver. 20.0 (IBM Co., Armonk, NY, USA).
Results
Baseline clinical characteristics and lab results were compared among the 3 groups (Table 1). The average age of patients was 9.8±2.4 years, and the average BA was 9.3±2.8 years. Twenty nine of the 113 participants achieved puberty on initiation of GH treatment, and all of them were in the ISS group. At baseline and after 12 months of GH treatment, there were no significant differences in Cobb angle and prevalence of scoliosis between prepubertal and pubertal subjects. Participants in the GHD group were younger and showed lower BA, IGF1-SDS, and IGFBP3-SDS than the ISS group. Also, both GHD and SGA groups showed lower height, height SDS, weight, and weight SDS than the ISS group. However, there were no significant differences in sex, BMI, BMI SDS, and thyroid function tests.
The average initial therapeutic GH dose was 0.29±0.04 mg/kg/wk, and the GHD group was treated with the lowest dose among the 3 groups, followed by the ISS and SGA groups in that order. Two of 26 GHD participants (7.7%), 1 of 9 SGA participants (10.3%), and 8 of 78 ISS participants (11.1%) had scoliosis at baseline.
Baseline clinical characteristics and lab results were compared after 12 months of GH treatment (Table 2). There was significant increase in height, weight, IGF1, IGF1-SDS, IGFBP3, and IGFBP3-SDS but no significant change in Cobb angle after GH treatment (6.2°±3.3° vs. 6.1°±3.5°, P=0.842).

Comparison of clinical characteristics, laboratory data and prevalence of scoliosis at baseline and 12 months after growth hormone treatment
Four of 11 subjects who were diagnosed with scoliosis at baseline still had scoliosis after treatment, and 7 subjects showed improvements with Cobb angles less than 10°. Eleven subjects were newly diagnosed with scoliosis after 12 months of GH treatment (Fig. 1). The prevalence of scoliosis was compared between baseline and 12 months after GH treatment (Table 2), and there was no significant difference (9.7% vs. 13.3%, P=0.481).

Illustration of subject groups during growth hormone treatment. GHD, growth hormone deficiency. SGA, small for gestational age. ISS, idiopathic short stature.
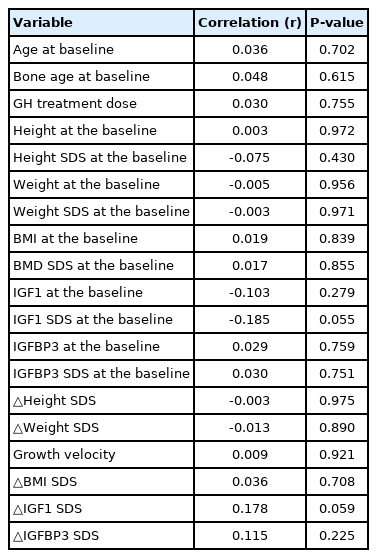
The correlations between Cobb angle at baseline, clinical characteristics, and lab data were analyzed, and Cobb angle had no significant correlation with any of the factors (Table 3). A change in Cobb angle during 12 months of treatment also had no statistically significant correlation with clinical characteristics or lab results (Table 4).
Discussion
This study found no significant change in the prevalence or worsening of scoliosis during GH treatment with increased height SDS, IGF1-SDS, and IGFBP3-SDS. Also, the average Cobb angles before and after 1 year of GH treatment were similar at 6.2°±3.3° and 6.1°±3.5°, respectively.
GH is an effective treatment for children with short stature, which is defined as a height below 2 standard deviations of the mean height of the general population matched for age, sex, and pubertal stage [17,24]. Side effects of GH therapy have been reported, ranging from a mild injection site reaction, headache, intracranial hypertension, slipped capital femoral epiphysis, to progression of scoliosis [25-28]. The incidence of these events is rare and varies by diagnostic group.
The effect of GH treatment on scoliosis is controversial. The baseline prevalence of scoliosis in children with GHD or ISS is similar to that in the general population (approximately 4%) but higher in children with PWS and Turner syndrome [14,15,29-31]. In the current study, the prevalence of scoliosis at baseline was 9.7%, higher than previously reported; however, the spontaneous regression rate (7 of 11, 63.6%) after a 1-year follow-up was also higher than those in previous reports (20%–30% of patients) [32,33]. In addition, since scoliosis is a 3-dimensional deformity of the spine, a sagittal x-ray view may be required for an accurate evaluation.34) However, in this study, only an AP x-ray view was performed, which may lead to overestimated or underestimated results. Also, some subjects may have been too young to assume correct postures, which could have led to misdiagnosis of scoliosis [34].
Several other studies have suggested that GH treatment does not increase the risk of scoliosis development and progression. Nakamura et al. reported that there was no difference in the prevalence of scoliosis between a GH treatment group and a nontreatment group in PWS [35]. Grootjen et al. [36] also reported that 8 years of GH treatment had no adverse effects on the prevalence and severity of scoliosis in children with PWS compared with age-matched untreated PWS children. Recently, Park et al. [15] analyzed 1,128 patients with ISS; the incidence of de novo scoliosis during GH treatment was 3.7%, similar to that in the general population.
In contrast, several studies have reported an increase in the prevalence and progression of scoliosis during GH treatment. Yun et al. [13] reported that the scoliosis measured by Cobb angle increased by 1 degree per year in patients who underwent GH treatment, while there was no significant annual change in the control group. Park et al. [15] reported that the progression rate of scoliosis was 16.4% during GH treatment, and the Cobb angle increased significantly by about 4 degrees compared to baseline during a mean of 5.5 months of GH treatment.
The pathophysiology of scoliosis remains largely unknown. Several studies have suggested its association with genetics, estrogen, melatonin, calmodulin, low bone mineral density, and vitamin D [37]. Additionally, the initiation and progression rates of scoliosis are high during pubertal growth spurts, suggesting the role of asynchronous neuro-osseous growth in scoliosis [38]. Ahl et al. [39] reported that the progression of scoliosis was observed during early puberty (stage 2) in girls with idiopathic scoliosis. In this stage, such girls have a higher endogenous secretion of GH than normal, implying an earlier growth spurt in scoliotic girls. Chung et al. [1] showed that peak serum luteinizing hormone level is positively correlated with the Cobb angle in central precocious puberty patients during gonadotropin-releasing hormone agonist treatment. However, there were no significant differences in Cobb angle and the prevalence of scoliosis between prepubertal and pubertal subjects in this study.
The progression of scoliosis during GH therapy seems to be associated with rapid growth rather than GH therapy itself [27]. The monitoring of IGF-1 and IGFBP-3 during treatment helps in the assessment of GH status in the investigation of short stature and contributes to the prediction of growth responses [40]. In the present study, height SDS increased significantly after treatment, indicating that GH treatment resulted in rapid growth, and there was significant increase in IGF1 and IGFBP3 levels after treatment.
A limitation of this study is its relatively small sample size and short duration. Second, there were no data on growth velocity before GH therapy; thus, changes in growth velocity were not evaluated. Third, there was no age- or sex-matched control group for comparison. Fourth, only an AP x-ray view was used to evaluate scoliosis. Even though the Cobb angle is the gold standard for diagnosis, the evaluation of only one x-ray view could have affected the results.
In conclusion, development or progression of scoliosis is a major concern in GH therapy. This study showed that the prevalence of scoliosis and Cobb angle did not change during 1 year of GH therapy, and height SDS increased. A long-term follow-up study with a larger cohort is required to better understand the natural course of scoliosis in GH-treated children.
Ethical statement
This study was approved by the Institutional Review Board (IRB) of Korea University Hospital. Informed consent was waived by the IRB (approval number: 2020AS0341).
Notes
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author contribution
Conceptualization: MP, YJK, KEO, EK, YR; Data curation: MP, YR; Formal analysis: MP, EK; Funding acquisition: YR; Methodology: MP, EK; Project administration: MP, YJK, KEO, EK, HN, YR, KL; Visualization: MP, YJ Kim, KE Oh, EK; Writing - original draft: MP; Writing - review & editing: MP, YJK, KEO, EK, HN, YR, KL