Vitamin D deficiency in children aged 6 to 12 years: single center's experience in Busan
Article information
Abstract
Purpose
This study investigated the prevalence and risk factors associated with vitamin D deficiency in children.
Methods
We analyzed the medical records of 330 patients from the age of 6 to 12, who visited the endocrinology clinic of the Department of Pediatrics at Pusan National University Hospital, from September, 2013 to May, 2014. According to their serum 25-hydroxyvitamin D (25(OH)D) levels, the patients were grouped into either the deficiency group (25(OH)D<20 ng/mL), or the sufficiency group (25(OH)D≥20 ng/mL). The differences between the 2 groups were compared.
Results
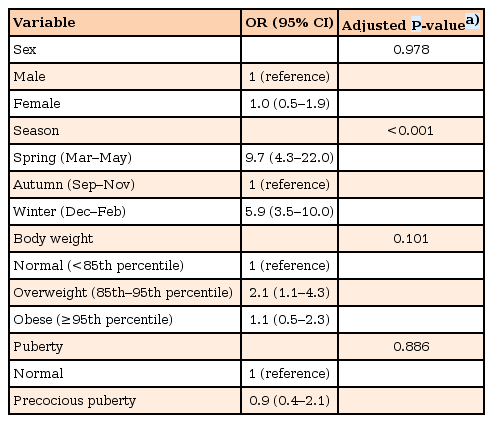
There were 195 patients (59.1%) who had vitamin D deficiency. Their mean serum 25(OH)D level was 14.86±3.20 ng/mL. The differences in sex, age, and pubertal status between the 2 groups were not statistically significant. Weight standard deviation score (SDS), and body mass index SDS, were significantly higher in the vitamin D deficiency group (P=0.002 for each), compared to the sufficiency group. Compared with Autumn, both Spring (odds ratio [OR], 9.7; 95% confidence interval [CI], 4.3–22.0), and Winter (OR, 5.9; 95% CI, 3.5–10.0), were risk factors for vitamin D deficiency. In multiple logistic regression analysis, only seasonal differences have been confirmed to have an effect on vitamin D deficiency.
Conclusion
Vitamin D deficiency in children aged 6 to 12 years is very common. Spring and Winter are the most important risk factors for vitamin D deficiency. We suggest that it is necessary to supplement the guideline for the vitamin D intake according to our situation.
Introduction
Vitamin D is an essential vitamin for the health and growth of bones. It is also important for calcium and phosphorus metabolism1). There are not many natural food items that contain vitamin D, therefore the amount of time in sunlight is the important source of vitamin D2). Vitamin D deficiency can result in skeletal diseases, such as rickets and osteomalacia3). In addition, vitamin D receptors exist in the endocrine glands and cardiovascular tissues, and take part in the differentiation of cell, and the production of various cytokines and interleukins, which, taken together, means that they are relevant in metabolic syndromes, diabetes, autoimmune diseases, and cardiovascular diseases456). Moreover, respiratory infections, food allergies, and asthma are known to increase with vitamin D deficiency, and there are also reports that menarche starts earlier in vitamin D deficient girls789). The known risk factors associated with vitamin D deficiency are skin pigmentation, older age, lesser sunlight exposure, lower intake of vitamin D rich food or supplements, and higher body mass index (BMI)51011).
Recently, vitamin D deficiency has become very common in both adults and children, due to a lack of exposure to sunlight12). There is still a controversy over the definition of vitamin D status131415). Therefore, the prevalence of vitamin D deficiency varies widely from country to country, ranging from 15 to 60 percent1016). As such, this study aimed to investigate the prevalence, and risk factors of vitamin D deficiency, in children aged 6 to 12 years, to provide a model of treatment.
Materials and methods
1. Subjects
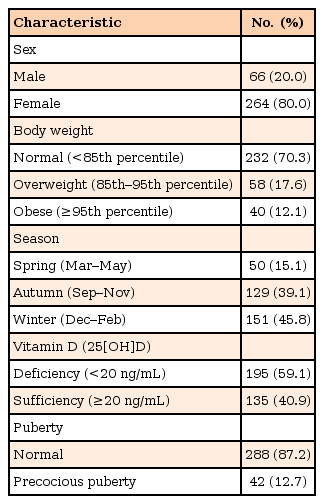
We targeted patients of 6 to 12 years of age, that visited the endocrinology clinic of the Department of Pediatrics at Pusan National University Hospital, from September, 2013 to May, 2014. We selected patients who visited our outpatient clinic for evaluation of growth and pubertal development without any acute illnesses on the day of visit. A total of 330 patients were included in this subject bracket (Table 1). The patients with chronic diseases, such as epilepsy and hypothyroidism, were excluded. Normal weight was defined as being in the 3rd to 84th percentile, in terms of BMI. Being overweight was defined as being in the 85th to 94th percentile. Obesity was defined as being in the 95th percentile or higher. This was based on the 2007 standard growth chart of Korean children and adolescents. There were 264 girls (80%) and 66 boys (20%). Among the patients, 232 (70.3%) had a normal weight, 58 (17.6%) were overweight, and 40 (12.1%) were obese (Table 1).
Central precocious puberty is the early activation of the hypothalamic-pituitary-gonadal axis, which occurs before 8 years of age in girls, and before 9 years of age in boys17). This study defined idiopathic central precocious puberty as the following: breast budding in girls before the age of 8, testicular volume equal to or more than 4 mL or 4 cm2 in boys before the age of 9, advanced bone age (1 year or more compared to the chronological age), and the peak luteinizing hormone level being equal to or higher than 5.0 IU/L on immunoradiometric assay following stimulation with gonadotropin-releasing hormone18). Of the total 330 patients, 42 (12.7%) were diagnosed with idiopathic central precocious puberty (Table 1).
2. Study design
We reviewed the participants' medical records to collect data on their height, weight, pubertal status, bone age, and serum levels of 25-hydroxyvitamin D (25(OH)D), calcium, phosphorus, and alkaline phosphatase (ALP). Pubertal status was evaluated according to the Tanner stages. Bone age was determined using the method defined by Greulich and Pyle. This study was approved by the Institutional Review Board of Pusan National University Hospital, Busan, Korea (approval number: E-2015071).
3. Laboratory measurements
The serum 25(OH)D levels were measured using an electrochemiluminescence binding assay (Roche Diagnostics, Indianapolis, IN, USA). The serum calcium, phosphorus, and ALP levels, were also measured. According to their serum 25(OH)D concentrations, the patients were divided into 2 groups; deficiency (25(OH)D<20 ng/mL), and sufficiency (25(OH)D≥20 ng/mL) groups. The definition of vitamin D status was based on the recommendation of the US Institute of Medicine (IOM) for vitamin D13).
4. Statistical analysis
SAS 9.3 (SAS Institute, Cary, NC, USA) was used for all statistical analysis. Continuous variables were expressed using the mean value and the associated standard deviation. The differences between the vitamin D deficiency group, and the sufficiency group, were compared using a 2-sample t-test. In terms of the categorical values, a chi-square test was used to compare the differences between the 2 groups. Multiple logistic regression analysis was used to analyze the factors that affected vitamin D deficiency. In all the statistical analyses, P-values below 0.05 were considered to be statistically significant.
Results
A total of 330 patients were included, and their characteristics are presented in Table 1. There were 195 patients (59.1%) in the vitamin D deficiency group, and 135 patients (40.9%) in the sufficiency group. The seasons that measurements were taken were Spring, with 50 patients (15.1%), Autumn, with 129 patients (39.1%), and Winter, with 151 patients (45.8%).
Table 2 shows the characteristics of the vitamin D deficiency and sufficiency groups. The mean serum levels of 25(OH)D were 14.86±3.20 ng/mL, and 27.01±5.59 ng/mL, respectively. The differences between the 2 groups in terms of sex, age, and pubertal status were not statistically significant. When the patients' weights were separated into normal, overweight, and obese, and the distribution was compared between the 2 groups, the differences were not statistically significant. However, the frequency of overweight and obese patients in the deficiency group was higher, compared to the sufficiency group. Weight standard deviation score (SDS), and BMI SDS, were significantly higher in the vitamin D deficiency group (P=0.002 for each), compared to the sufficiency group. There were 41 (82.0%) from the deficiency group, and 9 (18.0%) from the sufficiency group, in Spring. In Autumn, there were 42 (32.6%), and 87 (67.4%) and there were 112 (74.2%), and 39 (25.8%) from each group, respectively, in Winter. When compared with Autumn, the patients who were measured in Spring and Winter had higher percentage of vitamin D deficiency.
The mean serum calcium levels for the vitamin D deficiency group, and the sufficiency group, were 9.67±0.36 mg/dL and 9.76±0.32 mg/dL, respectively (Table 2), being significantly lower in the deficiency group (P=0.018). The mean serum phosphorus and ALP levels were not statistically different, between the 2 groups (Table 2). However, there was a tendency that ALP level was higher in the vitamin D deficiency group which had a lower calcium level. When multiple logistic regression was performed, to investigate the factors related to vitamin D deficiency, Spring (OR, 9.7; 95% CI, 4.3–22.0) and Winter (OR, 5.9; 95% CI, 3.5–10.0) were found to be factors significantly affecting vitamin D deficiency, when compared with Autumn (Table 3).
Discussion
In the present study, we found that vitamin D deficiency was very common among children aged 6 to 12 years. There were 195 patients (59.1%) with vitamin D deficiency, and their mean serum 25(OH)D level was 14.86±3.20 ng/mL. The mean serum 25(OH)D level of all subjects was 19.83±7.39 ng/mL which was also lower than the sufficient vitamin D concentration (≥20 ng/mL). The prevalence of vitamin D deficiency was found to increase in Spring and Winter, compared with Autumn.
In a previous study on the vitamin D status of Korean children, Lee et al.19) reported the mean serum level of 25(OH) D in 2,880 children and adolescents as 17.42±8.95 ng/mL. Another study reported that the prevalence of vitamin D deficiency (defined as 25(OH)D<20 ng/mL) in 1,212 children, aged between 4 and 15 years, was 58.6%20). These results are consistent with the findings of our study.
There is no consensus about the definition of vitamin D status for children and adolescents13141521). Globally, a serum 25(OH)D level below 10 ng/mL is considered the lowest cutoff for vitamin D status15). However, there are evidences that rickets can occur in infants and young children with a serum 25(OH)D concentration that is higher than 10 ng/mL322). The US IOM suggested that a serum 25(OH)D level above 20 ng/mL was sufficient for bone health13). After that, the Endocrine Society Task Force published a guideline that defined vitamin D sufficiency as a serum 25(OH)D>30 ng/mL, vitamin D insufficiency as a serum 25(OH)D 20–30 ng/mL and vitamin D deficiency as a serum 25(OH)D<20 ng/mL14). That was because there were increasing evidences that the optimal level of 25(OH)D might be above 30 ng/mL in adults2324). Recently, the Korean Nutrition Society used the definition of the IOM to set the dietary reference intake (DRI) for vitamin D in 201525).
It has been reported that the further away from the equator one resides, the shorter the yearly duration of ultraviolet B irradiation, sufficient for vitamin D synthesis26). In Korea (latitude 33–38°N), sunlight is known to be effective in synthesizing vitamin D from April to October, from 10 in the morning to 3 in the afternoon14). There are reports regarding the seasonal effect on variations in serum 25(OH)D levels according to differences in latitude. In the Northern hemisphere, serum 25(OH)D levels are reported to gradually decrease from Summer to Winter27282930). In the present study, the serum 25(OH)D levels measured in Spring and Winter, were lower than in Autumn, however Summer was not included in our study. According to Foo et al.31), the serum 25(OH)D levels of adolescent girls in Beijing, China (latitude 40°N), were 14.4 ng/mL when participating in sports, and 12 ng/mL when not participating in sports. Another study reported that during Winter, 40% of the adolescent girls tested, in Beijing, showed vitamin D deficiency. In Mongolia, (latitude 42–50°N), which is adjacent to China, ultraviolet light exposure is reported to be sufficient only in Summer, and the risk of vitamin D deficiency increases in Winter32).
The changes in lifestyle of modern society has caused an increase in indoor activities, decreasing exposure to sunlight among children and adolescents. Lee et al.33) reported that the time spent in the daylight was only 2.6 hr/wk in winter and 3.9 hr/wk in summer in 100 Korean children aged between 5 years and 13 years. Also they showed that vitamin D deficient children spent fewer hours outdoors in the daytime than vitamin D nondeficient children33). We did not measure the time spent in the sunlight, but the decrease in outdoor activities could also be a cause of the high prevalence of vitamin D deficiency in the present study. The Korean Nutrition Society determined the DRI for vitamin D under the assumption that sunlight exposure might be enough to meet vitamin D needs in Korean children25). The current DRI for vitamin D in Korean children aged 1–12 years is 200 IU/day which is lower than 600 IU/day of the IOM guideline13). We need more research on Korean children's sunlight exposure.
Obese individuals are known to have a lower level of vitamin D, compared with individuals of a normal weight34). In vitamin D deficiency, key enzymes that act on adipose tissue such as lipoprotein lipase and fatty acid synthase are thought to be inhibited, thereby affecting fat accumulation. Vitamin D3 is stored in subcutaneous fat, and the vitamin D3 synthesized in the skin of obese individuals become more sequestered. Due to this decrease in the bioavailability of vitamin D3, vitamin D deficiency appears more often in obese individuals35). The present study shows a significant difference between the vitamin D deficiency and sufficiency groups, in terms of weight SDS and BMI SDS. In spite of this, when multiple logistic regression analysis was used to analyze the factors affecting vitamin D deficiency, weight was not a statistically significant factor.
There have been several studies on the relationship between vitamin D status and sexual maturation. According to Grivas et al.36), menarche started at a younger age at Northern latitudes, where the level of vitamin D synthesis is lower. Villamor et al.37) followed a cohort of 242 healthy girls, from the age of 5 to 12 years, for 30 months, and observed that menarche occurred earlier in those with vitamin D deficiency. They explained that vitamin D deficiency is related to obesity and as such the vitamin D status would have indirectly affected menarche age. In the present study, there was no significant difference in pubertal status between the vitamin D deficiency and sufficiency groups.
There are a number of limitations to the present study. This is a hospital-based study, and most of the patients live in Busan, and as such this data may not be representative of the whole population of Korea. The levels of parathyroid hormone, which is one of the important factors affecting vitamin D status, were not measured. Of the 4 seasons, Summer was not included.
In conclusion, vitamin D deficiency in children aged 6 to 12 years is very common, and Spring and Winter are the most important risk factors for vitamin D deficiency. Efforts need to be made to improve the vitamin D status, such as increasing outdoor activities for more exposure to sunlight, and taking vitamin D supplements during seasons when vitamin D deficiency is widely prevalent. Furthermore, we suggest that it is necessary to supplement the guideline for vitamin D intake according to our situation.
Acknowledgments
This work was supported by a clinical research grant from Pusan National University Hospital 2015. We acknowledge assistance with statistical analysis from Pusan National University Hospital Clinical Trial Center Biostatistics Office.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.