Clinical and molecular review of atypical congenital adrenal hyperplasia
Article information
Abstract
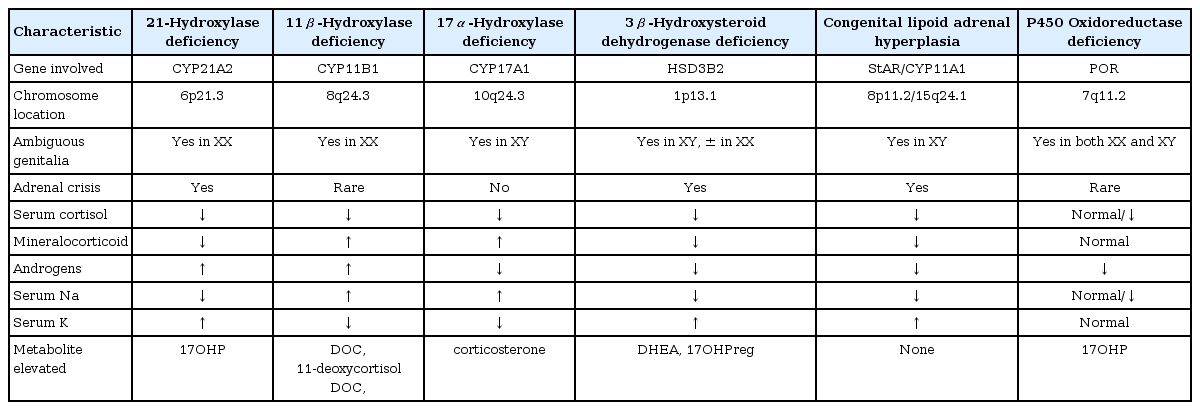
Congenital adrenal hyperplasia (CAH) is one of the most common inherited metabolic disorders. It comprises a group of autosomal recessive disorders caused by the mutations in the genes encoding for steroidogenic enzymes that involved cortisol synthesis. More than 90% of cases are caused by a defect in the enzyme 21-hydroxylase. Four other enzyme deficiencies (cholesterol side-chain cleavage, 17α-hydroxylase [P450c17], 11β-hydroxylase [P450c11β], 3β-hydroxysteroid dehydrogenase) in the steroid biosynthesis pathway, along with one cholesterol transport protein defect (steroidogenic acute regulatory protein), and one electrontransfer protein (P450 oxidoreductase) account for the remaining cases. The clinical symptoms of the different forms of CAH result from the particular hormones that are deficient and those that are produced in excess. A characteristic feature of CAH is genital ambiguity or disordered sex development, and most variants are associated with glucocorticoid deficiency. However, in the rare forms of CAH other than 21-hydroxylase deficiency so-called "atypical CAH", the clinical and hormonal phenotypes can be more complicated, and are not well recognized. This review will focus on the atypical forms of CAH, including the genetic analyses, and phenotypic correlates.
Introduction
Congenital adrenal hyperplasia (CAH) is one of the most common inherited metabolic disorders and is associated with significant morbidity and mortality in affected children. It comprises a group of autosomal recessive disorders caused by the mutations in the genes encoding for steroidogenic enzymes that involved cortisol synthesis. Impaired cortisol secretion results in hypersecretion of corticotropin-releasing hormone and adrenocorticotropic hormone (ACTH) and consequent hyperplasia of the adrenal glands. The clinical phenotypes and biochemical characteristics depend on the specific enzymatic defect. There is a broad clinical spectrum of this disorder. In most forms of CAH, it can be fatal if not diagnosed early in infancy1).
The adrenal cortex is the production site for three classes of steroid hormones: mineralocorticoids, glucocorticoids, and sex hormones. The cortex is divided into three zones by different cellular arrangements, each one functionally distinct due to the enzymes required for different hormone production. The outer zona glomerulosa does not express 17α-hydroxylase (P450c17)2) and hence produces 17-deoxysteroids leading to aldosterone (the most potent mineralocorticoid), and is regulated primarily by the renin/angiotensin system. The middle zona fasciculata expresses the 17α-hydroxylase activity but very little of the 17,20-lyase activity of P450c17, and hence produces 21-carbon, 17-hydroxysteroids, leading to cortisol under the influence of ACTH. The inner zona reticularis expresses both the 17α-hydroxylase and 17,20-lyase activities of P450c17, and hence produce the 19-carbon 17-hydroxy steroid dehydroepiandrosterone (DHEA), the precursor of sex steroids1,3).
ACTH regulates steroidogenesis (chronic regulation) by inducing the transcription of genes encoding various steroidogenic enzymes, but acute regulation is at the level of cholesterol access to cholesterol side-chain cleavage (P450scc). The steroidogenic acute regulatory protein (StAR) facilitates the movement of cholesterol into mitochondria, where it is converted to pregnenolone by P450scc4). Simplified diagram of adrenal steroidogenic pathways was shown in Fig. 1.
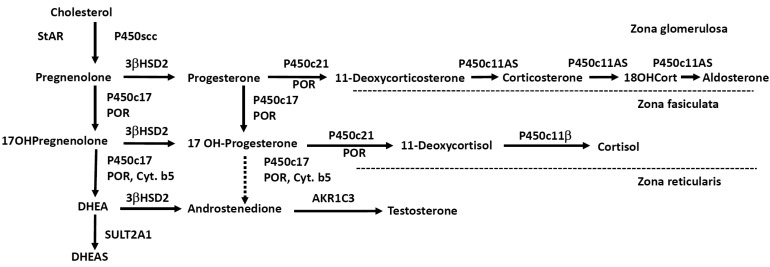
Simplified scheme of adrenal steroidogenesis. AKR, aldo-keto reductase; Cyt.b5, cytochrome b5; P450scc, cholesterol side-chain cleavage enzyme; P450c17, 17α-hydroxylase/17,20-lyase; 3βHSD2, 3β-hydroxysteroid dehydrogenase type 2; P450c21, 21-hydroxylase; P450c11AS, aldosterone synthase; P450c11β, 11β-hydroxylase; POR, P450 oxidoreductase; StAR, steroidogenic acute regulatory protein; SULT2A1, sulfotransferase; 18OHCort, 18-hydroxycorticosterone.
There have been nationwide newborn screening programs for CAH in only a few countries in Asia, although the recent pilot study suggested the high prevalence of CAH in Southeast Asian countries5,6). More than 90% of cases are caused by a defect in the enzyme 21-hydroxylase (P450c21). Four other enzyme deficiencies in the steroid biosynthesis pathway (P450scc, P450c17, 11β-hydroxylase [P450c11β], 3β-hydroxysteroid dehydrogenase [3βHSD]), along with one cholesterol transport protein defect (StAR), and one electron-transfer protein (P450 oxidoreductase; POR) account for the remaining cases. In these uncommon forms of CAH, the clinical and hormonal phenotypes can be complicated, and are not widely recognized by endocrinologists or pediatricians.
The clinical symptoms of the different forms of CAH result from the particular hormones that are deficient and those that are produced in excess. A characteristic feature of CAH is genital ambiguity or disordered sex development (DSD), and all variants are associated with glucocorticoid deficiency. Each variant of CAH is summarized in Table 1. In this review, we focus on the molecular genetic basis of the variant forms of CAH other than 21-hydroxylase so-called "atypical CAH", including the genetic analysis, and phenotypic correlates.
Congenital lipoid adrenal hyperplasia (lipoid CAH)
Congenital lipoid adrenal hyperplasia is the most severe form of CAH in which the synthesis of all adrenal and gonadal steroid hormones is impaired and leads to the accumulation of cholesterol esters7,8). The true incidence of lipoid CAH is unknown, but it is clearly much higher in Japanese, Korean and Palestinian population. Patients with classic lipoid CAH usually present with adrenal failure and salt wasting, beginning within the first few months of life, and have female external genitalia irrespective of genetic sex1).
Early clinical hormonal studies and incubations of affected tissue in vitro with various precursors identified a defect in the conversion of cholesterol to pregnenolone, so that the disorder was initially called "20,22 desmolase deficiency" as it was thought to result from a defect in the enzyme system converting cholesterol to pregnenolone; this enzyme was later identified as mitochondrial P450scc. However, in 1995 it was found that lipoid CAH results from mutations in the gene encoding the StAR7). StAR facilitates the movement of cholesterol into mitochondria, where it is converted to pregnenolone by P450scc4). StAR is expressed in the adrenals and gonads but not in the placenta9). Because placental production of progesterone is essential for the maintenance of human pregnancy, mutations in P450scc were thought to be incompatible with term gestation. Nevertheless, beginning in 2001 several patients with defects in CYP11A1 gene causing P450scc deficiency have been reported10,11,12,13,14,15,16,17). Most of these cases were caused by severe loss-of function mutations and presented with severe, early onset adrenal failure and complete phenotypic 46,XY sex reversal in genetic males14), although patients with late-onset nonclassic disease have been reported15,16,17,18). Thus, both of the defects of StAR and P450scc are now considered to be responsible for lipoid CAH, but most lipoid CAH may be caused by the mutations in the StAR gene.
More than 40 StAR mutations causing classic lipoid CAH have been described, but very few partial loss-of-function mutations have been reported19,20,21). The mutations are present in all exons. The mutations in intronic region are also found. The mutations causing premature translational termination or altering of the StAR reading frame are common, and they substantially alter the structure of the StAR protein. All missense mutations are found in the carboxy-terminal 40% of the amino acid StAR protein22). The Q258X mutation in exon 7 is very common in Japanese and Korean23,24). In Japanese this mutation is identified in 62% of the alleles and in over 80% of the patients. Other genetic clusters are found among Palestinian Arabs, most of whom carry the mutation R182L8); in eastern Saudi Arabia, carrying R182H25); and in parts of Switzerland, carrying the mutation L260P26).
Nonclassic lipoid CAH is a recently recognized disorder caused by StAR mutations that retain partial activity19). Affected individuals can present with later onset of adrenal insufficiency resembling nonautoimmune Addison disease with only mildly disordered sexual development or normal development with hypergonadotropic hypogonadism19,20,21). We recently reported four patients with nonclassic/atypical lipoid CAH and demonstrated that there is a broad clinical spectrum of StAR mutations21). While there is some variability in these biochemical assays, it seems that 10%-20% activity will dramatically alter the classic phenotype21). The R188C mutation was found in patients from Thailand, Canada, Jordan, India and Pakistan19,20,21), suggesting a recurrent mutation. To date, most patients with non-classic lipoid CAH carry R188C, although other mutations can cause this phenotype19,20,21).
3βHSD deficiency
3βHSD or Δ5→Δ4-isomerase is a 42 kDa microsomal enzyme catalyzes steroidogenic reactions: the conversion of the hydroxyl group to a keto group on carbon 3 and the isomerization of Δ5 steroids precursors into Δ4 ketosteroids27). Therefore, 3βHSD is responsible for the conversion of pregnenolone to progesterone, 17α-hydroxypregnenolone (17OHPreg) to 17α-hydroxyprogesterone (17OHP), DHEA to androstenedione, and androstenediol to testosterone. Thus, 3βHSD is an essential enzyme for biosynthesis of all classes of active steroid hormones including aldosterone, and cortisol in adrenal cortex, and sex steroids in adrenals and gonads.
In humans, there are two closely linked genes HSD3B1 and HSD3B2 located on chromosome 1 encoded two isoforms of 3βHSD28). The type 1 enzyme (3βHSD1) encoded by HSD3B1 is primarily expressed in placenta, mammary gland, liver, skin and some other tissues29). 3βHSD1 is required for placental progesterone synthesis during pregnancy. Mutations in HSD3B1 gene have never been described, presumably because these would cause a spontaneous abortion due to lack of placental progesterone synthesis. In contrast, the type 2 enzyme (3βHSD2) encoded by HSD3B2 gene is predominantly expressed in the adrenals and gonads29). Defects in HSD3B2 gene causes 3βHSD deficiency, which is a rare form of CAH, and can be fatal if not diagnosed early in infancy30).
The clinical spectrum of 3βHSD deficiency ranges from saltwasting to non-salt-wasting forms. In its classic form, 3βHSD deficiency causes various degrees of salt-wasting in both sexes. In genetic males, 3βHSD deficiency in the testes impairs testosterone biosynthesis from early fetal life, so that these males have undervirilization of the external genitalia, and usually present at birth with severe hypospadias and micropenis. By contrast, genetic females have normal female genitalia or slightly virilized genitalia such as isolated clitoromegaly, because the fetal adrenal overproduces large amounts of DHEA, which can be converted to testosterone by extraadrenal 3βHSD11,31). In this way, the presence of peripheral 3βHSD1 activity often complicates the hormonal diagnosis of this disorder in that very high 17OHPreg levels can be converted extra-adrenally to 17OHP confuses the diagnosis as 21-hydroxylase deficiency31,32).
Mild forms of 3βHSD deficiency cause premature acne, premature pubarche, and growth acceleration in children33) and a late onset variant manifesting with hirsutism, menstrual disorder, and polycystic ovaries in young women34,35). The newly proposed hormonal criteria for diagnosis for 3βHSD deficiency were elevated basal and ACTH-stimulated 17OHPreg and 17OHPreg to cortisol ratios, typically exceed 10 standard deviations above the mean36). These criteria were revised based on genotype-proven patients.
To date approximately 40 mutations have been identified in the HSD3B2 gene in patients suffering from classical 3βHSD deficiency. In most cases, the functional consequences of HSD3B2 mutations are in close agreement with the severity of the clinical manifestation. However, the in vitro 3βHSD activities alone cannot be used to predict the degree of male undervirilization31).
P450c11β deficiency
11β-hydroxylase deficiency (11OHD) accounts for about 5%-8% of CAH in people of European ancestry but accounts for about 15% of cases in both Muslim and Jewish Middle Eastern populations1). P450c11β catalyses conversion of 11-deoxycortisol to cortisol, representing the final step in cortisol biosynthesis. The enzyme also catalyses the monooxygenase reaction converting 11-deoxycorticosterone (DOC) to corticosterone. Thus, deficient P450c11β activity results in decreased cortisol secretion and accumulation of 11-deoxycortisol and the mineralocorticoid precursor DOC. Thus, patients can subsequently suffer from significant hypertension, a hallmark feature of this CAH variant. Accumulated precursors are shunted into the androgen synthesis pathway, leading to hyperandrogenism. Classic 11OHD most commonly results in 46,XX DSD with severe virilization of the external genitalia, and precocious pseudopuberty in both sexes. Newborns may also have elevated concentrations of 17OHP, which accumulates two steps behind the enzymatic block, so that P450c11β deficiency may be detected in newborn screening for P450c21 deficiency37). The diagnosis is established by elevated basal concentrations of DOC and 11-deoxycortisol, which hyperrespond to cosyntropin. 11OHD is caused by mutations in the CYP11B1 gene. At present, over 50 CYP11B1-inactivating mutations are described. Most are missense and nonsense mutations, but splice-site mutations, small deletions, small insertions, and complex rearrangements have also been detected38,39,40). The vast majority of mutations are associated with classic 11OHD, and only a few mutations causing nonclassic 11OHD have been described in otherwise asymptomatic women with hirsutism, and menstrual irregularities41,42).
Generally, the CYP11B1 gene is specifically amplified avoiding simultaneous amplification of homologous CYP11B2 sequences. In the majority of cases, molecular genetic analysis is not difficult. However, special cases are reported, such as an unequal crossing-over between the CYP11B2 and the CYP11B1 genes as a cause of 11OHD43).
P450c17 deficiency
P450c17 is the single microsomal cytochrome P450 enzyme that catalyzes both the 17α-hydroxylation required to produce the 17 hydroxy 21-carbon precursors of cortisol, 17OHPreg and 17OHP, and the 17,20-lyase activity needed to produce 19-carbon precursors of sex steroids44). P450c17 is encoded by CYP17A1 gene, consisting of eight exons and located on chromosome 10q24.345). CYP17A1 mutations cause P450c17 deficiency, a rare form of CAH characterized by sexual infantilism, 46,XY sex reversal, hypertension and high ratios of C21 to C19 steroids. The lack of P450c17 activity disrupts cortisol secretion, driving the compensatory overproduction of a glucocorticoid, corticosterone, and a mineralocorticoid, deoxycorticosterone, causing hypertension and hypokalemia1,46). Rare patients may also have isolated 17,20 lyase deficiency, characterized by low C19 steroids with normal cortisol47,48).
Over 70 CYP17A1-inactivating mutations have been identified. There is no evidence of a hot spot in most large populations. Therefore, sequencing of the entire coding region is usually necessary. Exceptions have been described in the some population, where mutations appear recurrently (1) a duplication of four nucleotides causing a frameshift is found among descendents of Dutch Frieslanders; (2) in-frame deletion of residues 487-489 is found throughout Southeast Asia; (3) a deletion of phenylalanine at position 53 or 54; and (4) the common W406R and R362C mutations, found among Brazilians of Spanish and Portuguese ancestry, respectively1).
Mutations underlying isolated 17,20 lyase deficiency result in amino acid substitutions located within the redox-partner binding site of P450c17, thereby disrupting the electron transfer from POR to P450c17 specifically for the conversion of 17OHPreg to DHEA47,48).
POR deficiency
POR deficiency is a unique and newly recognized form of CAH, biochemically manifesting with apparent combined P450c17 and P450c21 deficiency. POR transfers electrons from reduced nicotinamide adenine dinucleotide phosphate (NADPH) to all microsomal (type II) cytochrome P450 enzymes, including three steroidogenic enzymes: P450c17 (17α-hydroxylase/17,20 lyase), P450c21 (21-hydroxylase), and P450aro (aromatase)1). Although disruption of the POR gene in mice causes gross disorders of embryogenesis and embryonic lethality, in 2004 Fluck et al.49) reported POR mutations in three children with ambiguous genitalia and skeletal malformations (Antley-Bixler syndrome, ABS) and in a phenotypically normal adult woman with primary amenorrhea and polycystic ovaries. The majority of patients with POR deficiency described to date have also had the ABS phenotype, characterized by craniosynostosis, radioulnar or radiohumeral synostosis, bowed femora, and other variable skeletal disorders1,50). POR deficiency can cause ambiguous genitalia in both sexes. 46,XY males are typically undervirilized because decreased 17,20-lyase activity reduces androgen synthesis. 46,XX females are frequently virilized at birth, but this virilization is not progressive postnatally. There are two possible mechanisms for this virilization. First, because placental aromatase (P450aro) requires POR, a defect in this placental aromatase activity, either from mutation of POR or P450aro itself, will permit large amounts of fetal C19 steroids to enter and virilize the mother and the female fetus. Second, it appears to involve the "backdoor pathway" to fetal androgen production, in which 21-carbon steroid precursors are 5α-reduced and ultimately converted to dihydrotestosterone, bypassing the conventional precursors androstenedione and testosterone1,50).
The human POR gene consists of 16 exons, spanning approximately 70 kb on chromosome 7q11.2. The overall incidence of POR deficiency in the general population remains unclear. However, over 50 POR mutations have now been described, suggesting that this disorder may be relatively common. There is the great variability in the clinical and hormonal findings in POR deficiency. Some patients with milder POR mutations do not have ABS, and the steroidogenic defect may present as hypogonadism and/or infertility49,50,51,52). Two mutations are especially common: A287P, the predominant mutation in patients of European ancestry, and R457H, the predominant mutation in patients of Japanese ancestry53,54,55). The genotype-phenotype correlation is not fully established yet55,56), and future studies are needed.
Conclusions
There is a broad spectrum of the clinical and hormonal phenotypes of CAH depending on the specific enzymatic defect. The diagnosis remains a challenge in patients with atypical forms of CAH and requires thorough clinical and hormonal work-up. Lifelong treatment with steroids is required for most patients. Confirmation of the diagnosis by genetic analysis is of clinical importance.
Acknowledgments
The author is grateful to Drs. Walter L. Miller (University of California, San Francisco, CA, USA) and Vorasuk Shotelersuk (Chulalongkorn University, Bangkok, Thailand) for their excellent mentorship. T.S. was supported by Ratchadapiseksompotch funds from Faculty of Medicine, Chulalongkorn University, and the Thailand Research Fund.
Notes
Conflict of interest: No potential conflict of interest relevant to this article was reported.