Thyroid autoimmunity in children and adolescents with newly diagnosed type 1 diabetes mellitus
Article information
Abstract
Purpose
This study aim to investigate the occurrence of autoimmune thyroid disease in children and adolescents at onset of type 1 diabetes mellitus (T1DM) and to assess whether the presence of diabetes-specific autoantibodies can predict the autoimmune thyroid disorder.
Methods
Seventy-three children with T1DM were recruited. Glutamic acid decarboxylase antibodies (GADA), islet cell antibodies (ICA), insulin autoantibodies (IAA), and thyroid antibodies were determined in all patients at the time of diagnosis.
Results
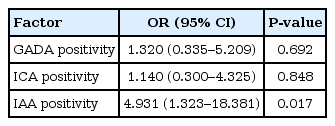
The majority of patients (87.7%) had at least one pancreatic antibody (74.0% for GADA, 20.5% for ICA, and 24.7% for IAA). Thyroid autoantibodies were found in 19 of 73 patients (26.0%) at diagnosis. Thyroid autoimmunity (TA) incidence was not statistically significant by GADA or ICA positivity, but significantly higher by IAA positivity (P=0.03), and IAA positivity showed odds ratio, 4.931; 95% confidence interval, 1.323-18.381 for TA.
Conclusion
The IAA positivity in children and adolescents with TIDM was strongly related to positivity of thyroid autoantibodies and thus it could serve as an index for early prediction of the development of the thyroid autoimmune disorder among children and adolescents with TIDM.
Introduction
Type 1 diabetes mellitus (T1DM) is a common endocrine disorder associated with aberrant immune responses to specific β-cell autoantigens1,2). Markers of the process are various including autoantibodies to glutamic acid decarboxylase (GADA), to islet cell (ICA) and to insulin (IAA)3). These patients are also prone to other autoimmune disorders such as Graves' disease, Hashimoto's thyroiditis, Addison's disease, vitiligo, celiac sprue, autoimmune hepatitis, myasthenia gravis, and pernicious anemia4).
The most prevalent autoimmune disease associated with T1DM is autoimmune thyroid disorder, characterized by the presence of thyroid antibodies to peroxidase and thyroglobulin. The prevalence of positive thyroid antibodies in children with T1DM is ~50% in different countries but varies with age, sex, and ethnic origin of patients5). Thyroid diseases occur within 3 to 4 years in half of the thyroid antibody positive groups6,7). It is inferred that TIDM and autoimmune thyroiditis are based on a common genetic origin, as they have similar pathogenicity and frequently occur simultaneously in the same individual and/or the same family8). Some pancreatic antibodies, known to be related to autoimmune diseases, are specially associated with an autoimmune reaction to thyroid antigens9,10).
The aim of the present study was to investigate the occurrence of autoimmune thyroid disease in children and adolescents at the onset of T1DM and to assess whether the presence of diabetes-specific autoantibodies can predict thyroid autoimmunity (TA).
Materials and methods
1. Subjects
The study was undertaken at the Chonnam National University Hospital and Chonbuk National University Hospital located in the Honam region of Korea. We retrospectively analyzed the medical records from children and adolescents who had been diagnosed with T1DM between January 2007 and June 2011. All patients met the criteria of American Diabetes Association for TIDM4). Patients who were not tested for all pancreatic autoantibodies and thyroid antibodies were excluded. We reviewed the demographic data, history of diabetes onset, and thyroid palpation findings. This study was approved by the Institutional Review Board of each hospital. The recommendations of the Declaration of Helsinki for biomedical research involving human subjects were followed.
2. Methods
Levels of blood glucose and glycosylated hemoglobin (HbA1c) were assessed and GADA, ICA, and IAA were evaluated at the time of diagnosis. Screening for TA was performed using measurements of thyroid antiperoxidase antibody (TPOAb), antithyroglobulin antibody (TGAb), and thyroid stimulating hormone (TSH) receptor antibodies (TSHRAb). Free thyroxine and TSH were also measured. TA was defined as the presence of at least one thyroid autoantibody. GADA was assessed by radioimmunoassay kit (RSR Ltd., Cardiff, UK), IAA by enzyme immunoassay kit (Orgentec Diagnostika, Mainz, Germany), and ICA by the indirect fluorescent antibody technique (SCIMEDX, Denville, NJ, USA). TPOAb (normal range, 0-34 IU/mL) and TGAb (normal range, 0-115 IU/mL) were measured using electrochemiluminiscence immunoassay kit (COBAS, Roche Diagnostics GmbH, Mannheim, Germany) and TSHRAb (normal range, 0-15 IU/L) was determined by radioimmunoassay (RSR Ltd.).
3. Statistical analysis
Data are expressed as mean±standard deviation or proportion (%). The unpaired Student t-test was performed to compare continuous variables, and the chi-square test was used to compare percentages among different patient subgroups. Multiple logistic regression analysis was used to assess the strength and independency of the associations. Outcomes included pancreatic antibody positivity and TA. All statistical analyses were performed with IBM SPSS ver. 20.0 (IBM Co., Armonk, NY, USA). A P-value <0.05 was considered significant.
Results
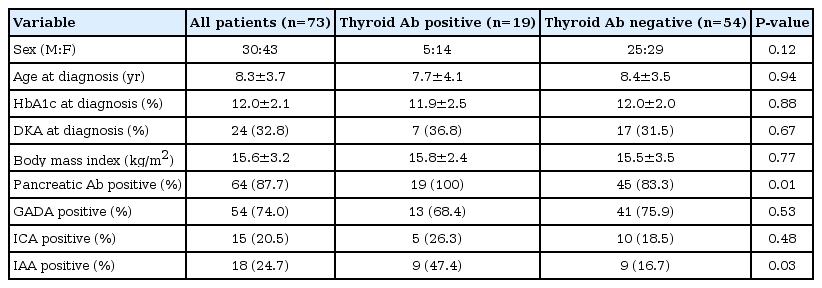
Of the 98 patients selected for the study, 25 patients who were not tested for all pancreatic autoantibodies and thyroid antibodies were excluded. Therefore, 73 patients were studied. Of the 73 patients, 30 were boys (41.1%) and 43 (58.9%) were girls. The mean age at diagnosis of diabetes was 8.3±3.7 years. Mean HbA1c at onset of diabetes was 12.0%±2.1% and body mass index was 15.6±3.2 kg/m2. The majority of patients (64 of 73, 87.7%) had at least one pancreatic antibody at onset of TIDM, whereas only nine patients (12.3%) were negative for all three antibodies. In particular, 54 patients (74.0%) were positive for GADA, 15 (20.5%) for ICA, and 18 (24.7%) for IAA, respectively (Table 1). Only one antibody was present in 44 patients (60.3%), 17 patients (23.3%) were positive for two of three, and three (4%) for all three antibodies. Nineteen patients (26.0%) were positive for thyroid autoantibody at diagnosis of TIDM. 73.7% of patients with positive thyroid antibodies were female compared with 53.7% in patients without antibodies.
All 19 patients in the thyroid antibody positive group were positive for at least one pancreatic autoantibody (100% vs. 83.3%, P=0.01) (Table 1). Of the 19 patients with TA, 13 (68.4%) patients were positive for GADA, 5 (26.3%) were positive for ICA. And nine of 19 (47.4%) were significantly positive for IAA (P=0.03). The prevalence of TPOAb, TGAb, and TSHRAb was 15.1%, 21.9%, and 2.7% in patients with T1DM, respectively. Only 1 antibody was present in 10 patients (13.7%), eight patients (11.0%) were positive for two of three, and one (1.3%) for all three antibodies.
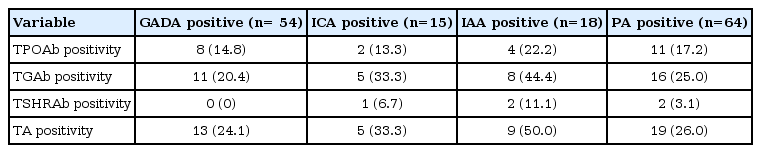
The prevalence rates of thyroid autoantibodies are shown according to the presence of each pancreatic antibody (Table 2). The prevalence of overall TA was as follows; 13/54 (24.1%) in the GADA positive group, 5/15 (33.3%) in the ICA positive group, and 9/18 (50%) in the IAA positive group, respectively. The prevalence of TA was not significantly influenced by GADA or ICA positivity, but was significantly increased by IAA positivity (odds ratio, 4.931; 95% confidence interval, 1.323-18.381) (Table 3).
Discussion
T1DM is an autoimmune disease in most cases. The incidence of T1DM varies widely across the world, from the highest incidence in northern Europe to the lowest incidence in Asia11). T1DM is frequently associated with other autoimmune disorders, particularly endocrinological diseases7,8,12,13). Autoimmune thyroid disorders are the most prevalent immunological diseases in patients with T1DM7,8,12). Autoimmune destruction of the thyroid gland may cause either hypothyroidism or hyperthyroidism. We identified autoantibodies to islet cells and the thyroid in a number of Korean children and adolescents with TIDM. Substantial variations in the prevalence of autoantibodies have been reported in other ethnic groups. The prevalence rates of antibodies in Japanese patients with TIDM are 60%-70% for GADA, 45%-50% for IAA, and 60%-65% for IA-2 autoantibodies at disease onset, which are similar to those reported in Caucasian patients14). In our study, 64 patients (87.7%) had at least one pancreatic antibody (GADA, 74.0%; ICA, 20.5%; and IAA, 24.7%).
The prevalence of thyroid dysfunction increases with the presence of thyroid autoantibodies. TPOAb and/or TGAb can be detected long before changes in TSH and thyroid hormone levels occur5,7,8). Thus determining these antibody levels might be useful for early diagnosis of the disease before thyroid dysfunction develops. The prevalence of thyroid autoantibodies in children with T1DM varies widely between 3% and 50% according to different studies5,7,8,13,15). It has been suggested that the coexistence of thyroid dysfunction might cause disturbances in glycemic control of patients with diabetes16). Even subclinical hypothyroidism (slightly elevated TSH without impairment of T4 and T3 levels) is associated with a higher frequency of symptomatic hypoglycemia17). Furthermore, Chase et al.18) reported a reduced rate of growth in diabetic children with elevated TSH values and thyromegaly, but normal serum thyroxine levels. Treatment with L-thyroxine leads to significantly improved growth in prepubertal children compared with age-matched diabetic controls. Thyroid function tests and ultrasound assessments are recommended in cases of antibody positivity to minimize the risk of undiagnosed hypothyroidism in young patients with diabetes. From our study, the prevalence rates of TPOAb, TGAb, and TSHRAb were 15.1%, 21.9%, and 2.7% in patients with T1DM, respectively. The overall prevalence of TA was 26.0%, which was similar to the findings in other studies. In Germany and Austria, Kordonouri et al.5,10) reported a prevalence of 21% TA, whereas a prevalence of 50% has been reported in a Caucasian population with diabetes in the USA6). Other Asian studies have reported a prevalence of 21.8% in a Taiwanese population and 18% in a Japanese population19,20).
Jin et al.9) found that thyroid antibody-positive TIDM patients have a higher frequency of GADA than thyroid antibody-negative patients and that a high titer of GADA is a strong predictor for the development of TA in patients with TIDM. A possible explanation for this association could be that GAD is not exclusively present in the pancreas but can also be found in other tissues, such as the follicle cells of the thyroid gland and the parietal cells of the stomach21,22). Thus, the persistence of GADA could be a marker for the future development of autoimmunity against the thyroid gland and other organs. Chang et al.19) reported the absence of correlation between TPOAb and GADA in Taiwanese TIDM patients and suggested genetic heterogeneity in the role of autoimmunity of TIDM and autoimmune thyroid disease among races. In the present study, the prevalence of overall thyroid antibody positivity was as follows; 13 (24.1%) in the GADA-positive group, five (33.3%) in the ICA-positive group, and nine (50%) in the IAA positive group, respectively. TA incidence was not statistically significant by GADA or ICA positivity but significantly higher by IAA positivity, and IAA positivity showed a odds ratio, 4.931; 95% confidence interval, 1.323-18.381 for TA.
In conclusion, the prevalence of TA in our study was 26.0% in children and adolescents with TIDM. IAA positivity in children and adolescents with TIDM was strongly related to the positivity of thyroid autoantibodies and thus it could serve as an index for early prediction of the development of the thyroid autoimmune disorder among children and adolescents with TIDM. Long-term prospective investigations with large populations of individuals are needed to elucidate the natural history of autoimmunity in patients with TIDM.
Notes
No potential conflict of interest relevant to this article was reported.